Β-Pinene
Chemical compound From Wikipedia, the free encyclopedia
Chemical compound From Wikipedia, the free encyclopedia
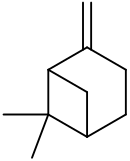
β-Pinene is a monoterpene, an organic compound found in plants. It is one of the two isomers of pinene, the other being α-pinene. It is a colorless liquid soluble in alcohol, but not water. It has a woody-green pine-like smell.
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
6,6-Dimethyl-2-methylidenebicyclo[3.1.1]heptane Pin-2(10)-ene | |||
| Other names
6,6-Dimethyl-2-methylenebicyclo[3.1.1]heptane 2(10)-Pinene Nopinene Pseudopinene | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.004.430 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C10H16 | |||
| Molar mass | 136.238 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 0.872 g/mL | ||
| Melting point | −61.54 °C; −78.77 °F; 211.61 K[1] | ||
| Boiling point | 165–167 °C; 329–332 °F; 438–440 K[2] | ||
| Thermochemistry | |||
Std enthalpy of combustion (ΔcH⦵298) |
−6214.1±2.9 kJ/mol[1] | ||
| Hazards | |||
| GHS labelling: | |||
    | |||
| Danger | |||
| H226, H304, H315, H317, H410 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P272, P273, P280, P301+P310, P302+P352, P303+P361+P353, P321, P331, P332+P313, P333+P313, P362, P363, P370+P378, P391, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 36 °C (97 °F; 309 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
β-Pinene is one of the most abundant compounds released by forest trees.[3] If oxidized in air, the allylic products of the pinocarveol and myrtenol family prevail.[4]
Many plants from many botanical families contain the compound, including:
The clear compound is produced by distillation of turpentine oils.[10]
This section needs expansion. You can help by adding to it. (October 2023) |
β-Pinene is used in fragrances and essential oils. It is also used in the production of other aroma compounds, such as myrcene and nerol (got by careful fractional distillation of crude nerol got from myrcene[11]). The myrcene is got by pyrolysis of α-Pinene or β-Pinene.[12] Reacting with formaldehyde, result is nopol. When nopol is acetylated, the result is nopyl acetate, which is used as fragrance material.[10][13]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.