
Adrenochrome
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Adrenochrome?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Quick Facts Names, Identifiers ...
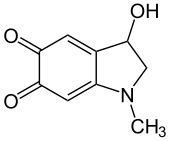 | |
 | |
| Names | |
|---|---|
| IUPAC name
3-Hydroxy-1-methyl-2,3-dihydro-1H-indole-5,6-dione | |
| Other names
Adraxone; Pink adrenaline | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.176 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
| Properties | |
| C9H9NO3 | |
| Molar mass | 179.175 g·mol−1 |
| Appearance | deep-violet[1] |
| Density | 3.264 g/cm3 |
| Boiling point | 115–120 °C (239–248 °F; 388–393 K) (decomposes) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
This article is about the chemical compound. For the conspiracy theory about Satanic child sacrifice, see QAnon.
For the Sisters of Mercy song, see Some Girls Wander by Mistake.
Adrenochrome is a chemical compound produced by the oxidation of adrenaline (epinephrine). It was the subject of limited research from the 1950s through to the 1970s as a potential cause of schizophrenia. While it has no current medical application, the semicarbazide derivative, carbazochrome, is a hemostatic medication.
Despite this compound's name, it is unrelated to the element chromium; instead, the ‑chrome suffix indicates a relationship to color, as pure adrenochrome has a deep violet coloration.[1]