Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
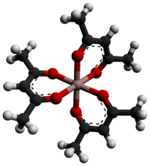
Aluminium acetylacetonate, also referred to as Al(acac)3, is a coordination complex with formula Al(C5H7O2)3. This aluminium complex with three acetylacetone ligands is used in research on Al-containing materials. The molecule has D3 symmetry, being isomorphous with other octahedral tris(acetylacetonate)s.[2]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tris(acetylacetonato)aluminium | |
| Other names
Aluminium acetylacetonate, aluminium acetylacetonate | |
| Identifiers | |
| ECHA InfoCard | 100.034.296 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| Al(C5H7O2)3 | |
| Molar mass | 324.31 g/mol |
| Appearance | White solid[1] |
| Density | 1.42 g/cm3 |
| Melting point | 190-193 °C |
| Boiling point | 315 °C |
| Low | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aluminium acetylacetonate can be used as the precursor to crystalline aluminium oxide films using low-pressure metal organic chemical vapour deposition.[3] In horticulture it can also be used as a molluscicide.[4]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.