
Bismuth telluride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Bismuth telluride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Bismuth telluride (Bi2Te3) is a gray powder that is a compound of bismuth and tellurium also known as bismuth(III) telluride. It is a semiconductor, which, when alloyed with antimony or selenium, is an efficient thermoelectric material for refrigeration or portable power generation. Bi2Te3 is a topological insulator, and thus exhibits thickness-dependent physical properties.
Quick Facts Identifiers, Properties ...
 Single crystal of bismuth telluride | |
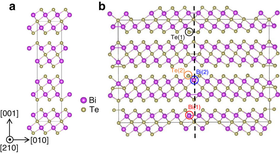 Atomic structure: ideal (l) and with a twin defect (r) | |
 Electron micrograph of twinned bismuth telluride | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.013.760 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Bi2Te3 | |
| Molar mass | 800.76 g·mol−1 |
| Appearance | Grey powder or metallic grey crystals |
| Density | 7.74 g/cm3[1] |
| Melting point | 580 °C (1,076 °F; 853 K)[1] |
| insoluble[1] | |
| Solubility in ethanol | soluble[1] |
| Structure | |
| Trigonal, hR15 | |
| R3m, No. 166[2] | |
a = 0.4395 nm, c = 3.044 nm | |
Formula units (Z) |
3 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | noncombustible[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp) (pure) none (doped with selenium sulfide) [3] |
REL (Recommended) |
TWA 10 mg/m3 (total) TWA 5 mg/m3 (resp) (pure) TWA 5 mg/m3 (doped with selenium sulfide)[3] |
IDLH (Immediate danger) |
N.D. (pure and doped)[3] |
| Safety data sheet (SDS) | Sigma-Aldrich |
| Related compounds | |
Other anions |
|
Other cations |
Arsenic telluride Antimony telluride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
