
Caesium iodide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cesium iodide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"CsI" redirects here. For other uses, see CSI (disambiguation).
Caesium iodide or cesium iodide (chemical formula CsI) is the ionic compound of caesium and iodine. It is often used as the input phosphor of an X-ray image intensifier tube found in fluoroscopy equipment. Caesium iodide photocathodes are highly efficient at extreme ultraviolet wavelengths.[7]
Quick Facts Names, Identifiers ...
 CsI crystal | |
 Scintillating CsI crystal | |
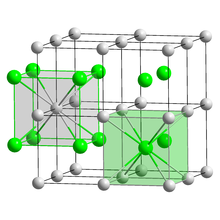 Crystal structure | |
 | |
| Names | |
|---|---|
| IUPAC name
Caesium iodide | |
| Other names
Cesium iodide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.029.223 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CsI | |
| Molar mass | 259.809 g/mol[2] |
| Appearance | white crystalline solid |
| Density | 4.51 g/cm3[2] |
| Melting point | 632 °C (1,170 °F; 905 K)[2] |
| Boiling point | 1,280 °C (2,340 °F; 1,550 K)[2] |
| 848 g/L (25 °C)[2] | |
| -82.6·10−6 cm3/mol[3] | |
Refractive index (nD) |
1.9790 (0.3 µm) 1.7873 (0.59 µm) 1.7694 (0.75 µm) 1.7576 (1 µm) 1.7428 (5 µm) 1.7280 (20 µm)[4] |
| Structure | |
| CsCl, cP2 | |
| Pm3m, No. 221[5] | |
a = 0.4503 nm | |
Lattice volume (V) |
0.0913 nm3 |
Formula units (Z) |
1 |
| Cubic (Cs+) Cubic (I−) | |
| Thermochemistry | |
Heat capacity (C) |
52.8 J/mol·K[6] |
Std molar entropy (S⦵298) |
123.1 J/mol·K[6] |
Std enthalpy of formation (ΔfH⦵298) |
−346.6 kJ/mol[6] |
Gibbs free energy (ΔfG⦵) |
-340.6 kJ/mol[6] |
| Hazards | |
| GHS labelling: | |
   | |
| Warning | |
| H315, H317, H319, H335 | |
| P201, P202, P261, P264, P270, P271, P272, P273, P280, P281, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P321, P330, P332+P313, P333+P313, P337+P313, P362, P363, P391, P403+P233, P405, P501 | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
2386 mg/kg (oral, rat)[1] |
| Related compounds | |
Other anions |
Caesium fluoride Caesium chloride Caesium bromide Caesium astatide |
Other cations |
Lithium iodide Sodium iodide Potassium iodide Rubidium iodide Francium iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close