Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
Clozapine N-oxide (CNO) is a synthetic drug used mainly in biomedical research as a ligand to activate DREADD receptors.[1] Although CNO was initially believed to be biologically inert. However, it has been shown not to enter the brain after administration[2] and to reverse metabolise in peripheral tissues to form clozapine. Clozapine can bind to a number of different serotonergic, dopaminergic and adrenergic receptors within the brain.[3] Therefore, behavioural data using the CNO-DREADD system in neuroscience experiments have to be interpreted with caution.[4]
 | |
| Names | |
|---|---|
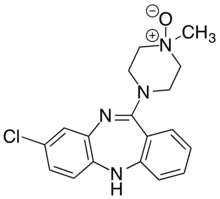
| IUPAC name
3-chloro-6-(4-methyl-4-oxidopiperazin-4-ium-1-yl)-11H-benzo[b][1,4]benzodiazepine | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.164.243 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C18H19ClN4O | |
| Molar mass | 342.83 g·mol−1 |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H301, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
This article may be too technical for most readers to understand. (March 2021) |
Alternatives to CNO with more affinity, more inert character, and faster kinetics include Compound 21 (C21)[5] and deschloroclozapine (DCZ).[6]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.