
Creatine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Creatine monohydrate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with creatinine or keratin.
Creatine (/ˈkriːətiːn/ or /ˈkriːətɪn/)[1] is an organic compound with the nominal formula (H2N)(HN)CN(CH3)CH2CO2H. It exists in various tautomers in solutions (among which are neutral form and various zwitterionic forms). Creatine is found in vertebrates where it facilitates recycling of adenosine triphosphate (ATP), primarily in muscle and brain tissue. Recycling is achieved by converting adenosine diphosphate (ADP) back to ATP via donation of phosphate groups. Creatine also acts as a buffer.[2]
Quick Facts Names, Identifiers ...
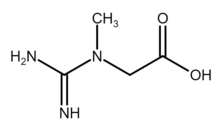 Skeletal formula of neutral form of creatine | |
 Skeletal formula of one of the zwitterionic forms of creatine | |
 Ball and stick model of one zwitterionic form of creatine | |
| Names | |
|---|---|
| Systematic IUPAC name
2-[Carbamimidoyl(methyl)amino]acetic acid | |
| Other names
N-Carbamimidoyl-N-methylglycine; Methylguanidoacetic acid; N-amidinosarcosine | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3DMet | |
| 907175 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.000.278 |
| EC Number |
|
| 240513 | |
| KEGG |
|
| MeSH | Creatine |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H9N3O2 | |
| Molar mass | 131.135 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| Melting point | 255 °C (491 °F; 528 K) |
| 13.3 g L−1 (at 18 °C) | |
| log P | −1.258 |
| Acidity (pKa) | 3.429 |
| Basicity (pKb) | 10.568 |
| Isoelectric point | 8.47 |
| Thermochemistry | |
Heat capacity (C) |
171.1 J K−1 mol−1 (at 23.2 °C) |
Std molar entropy (S⦵298) |
189.5 J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) |
−538.06–−536.30 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−2.3239–−2.3223 MJ mol−1 |
| Pharmacology | |
| C01EB06 (WHO) | |
| Pharmacokinetics: | |
| 3 hours | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P305+P351+P338 | |
| Related compounds | |
Related alkanoic acids |
|
Related compounds |
Dimethylacetamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close