
Desmosterol
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Desmosterol?
Summarize this article for a 10 year old
Desmosterol (Cholesta-5,24-dien-3β-ol) is a lipid present in the membrane of phytoplankton and an intermediate product in cholesterol synthesis in mammal cells.[1] Structurally, desmosterol has a similar backbone to cholesterol, with the exception of an additional double bond in the structure of desmosterol.
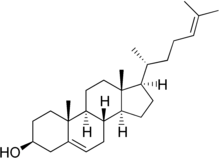 | |
 | |
| Names | |
|---|---|
| IUPAC name
Cholesta-5,24-dien-3β-ol | |
| Systematic IUPAC name
(1R,3aS,3bS,7S,9aR,9bS,11aR)-9a,11a-Dimethyl-1-[(2R)-6-methylhept-5-en-2-yl]-2,3,3a,3b,4,6,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-cyclopenta[a]phenanthren-7-ol | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.005.671 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H44O | |
| Molar mass | 384.64 g/mol |
| Appearance | White powder |
| Melting point | 121.5 °C (250.7 °F; 394.6 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The similarity can be seen biologically through the synthesis of cholesterol in the human body, as desmosterol is the immediate precursor to cholesterol in the Bloch pathway.[2] Desmosterol is accumulated in desmosterolosis and undergoes reduction with the catalyst 24-dehydrocholesterol reductase to form cholesterol.[3]
In 2014, desmosterol was named the Molecule of the Year 2012 by the International Society for Molecular and Cell Biology and Biotechnology Protocols and Researches (ISMCBBPR).[4]
