Dihydroxymalonic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dihydroxymalonic acid?
Summarize this article for a 10 year old
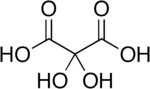
Dihydroxymalonic acid is an organic compound with formula C3H4O6 or HO-(C=O)-C(OH)2-(C=O)-OH, found in some plants such as alfalfa and in beet molasses.[2]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Dihydroxypropanedioic acid | |
| Other names
Mesoxalic acid monohydrate Oxomalonic acid monohydrate Ketomalonic acid monohydrate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.372 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H4O6 | |
| Molar mass | 136.059 g·mol−1 |
| Melting point | 119 to 120 °C (246 to 248 °F; 392 to 393 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound is also called dihydroxymesoxalic acid and dihydroxypropanedioic acid. It can be viewed as a hydrate derivative of mesoxalic acid, and is often called mesoxalic acid monohydrate and similar names.[3] This compound is unusual in containing stable geminal hydroxy groups.
Dihydroxymalonic acid is a water-soluble white solid. It crystallizes in deliquescent prisms that melt between 113 °C and 121 °C without loss of water.[4] It has been used in medical research as a hypoglycemic agent[5] and was patented in the United States in 1997 as a fast-acting antidote to cyanide poisoning.[6]