
Dimethyl sulfide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dimethylsulfide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
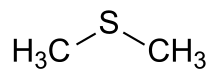
Dimethyl sulfide (DMS) or methylthiomethane is an organosulfur compound with the formula (CH3)2S. The simplest thioether, it is a flammable liquid that boils at 37 °C (99 °F) and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize, cabbage, beetroot, and seafoods. It is also an indication of bacterial contamination in malt production and brewing. It is a breakdown product of dimethylsulfoniopropionate (DMSP), and is also produced by the bacterial metabolism of methanethiol.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(Methylsulfanyl)methane[3] | |
| Other names | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3DMet | |
| 1696847 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.000.770 |
| EC Number |
|
| KEGG |
|
| MeSH | dimethyl+sulfide |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1164 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| (CH3)2S | |
| Molar mass | 62.13 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Stench: cabbage, sulfurous, unpleasant |
| Density | 0.846 g·cm−3 |
| Melting point | −98 °C; −145 °F; 175 K |
| Boiling point | 35 to 41 °C; 95 to 106 °F; 308 to 314 K |
| log P | 0.977 |
| Vapor pressure | 53.7 kPa (at 20 °C) |
| −44.9×10−6 cm3/mol | |
Refractive index (nD) |
1.435 |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−63.9 to −66.9 kJ⋅mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−2.1812 to −2.1818 MJ⋅mol−1 |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H225, H315, H318, H335 | |
| P210, P261, P280, P305+P351+P338 | |
| Flash point | −36 °C (−33 °F; 237 K) |
| 206 °C (403 °F; 479 K) | |
| Explosive limits | 19.7%[clarification needed] |
| Safety data sheet (SDS) | osha.gov |
| Related compounds | |
Related chalcogenides |
|
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close