
Ebastine
Antihistamine drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ebastine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Ebastine is a H1 antihistamine with low potential for causing drowsiness.
"Aleva" redirects here. For the pepper called "aleva", see Cayenne pepper.
Quick Facts Clinical data, AHFS/Drugs.com ...
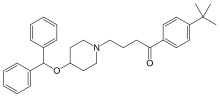 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | Greater than 95% |
| Metabolism | Hepatic (CYP3A4-mediated), to carebastine |
| Elimination half-life | 15 to 19 hours (carebastine) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.106.831 |
| Chemical and physical data | |
| Formula | C32H39NO2 |
| Molar mass | 469.669 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Close
It does not penetrate the blood–brain barrier to a significant amount and thus combines an effective block of the H1 receptor in peripheral tissue with a low incidence of central side effects, i.e. seldom causing sedation or drowsiness.[1][2][3]
It was patented in 1983 by Almirall S.A and came into medical use in 1990.[4] The substance is often provided in micronised form due to poor water solubility.