
Eplivanserin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Eplivanserin?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
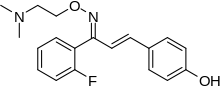
Eplivanserin (SR-46,349; planned trade name Ciltyri) was an experimental drug for the treatment of insomnia which was being developed by Sanofi Aventis.[1]
Quick Facts Clinical data, Routes ofadministration ...
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ECHA InfoCard | 100.189.857 |
| Chemical and physical data | |
| Formula | C19H21FN2O2 |
| Molar mass | 328.387 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Close
Sanofi Aventis announced in December 2009 that it was withdrawing its application for approval of eplivanserin from both the U.S. Food and Drug Administration and the European Medicines Agency.[2]