
Tetrathiafulvalene
Organosulfuric compound with formula C6H4S4 / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Fabre salt?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Tetrathiafulvalene (TTF) is an organosulfur compound with the formula (C3H2S2)2. Studies on this heterocyclic compound contributed to the development of molecular electronics. TTF is related to the hydrocarbon fulvalene, (C5H4)2, by replacement of four CH groups with sulfur atoms. Over 10,000 scientific publications discuss TTF and its derivatives.[2]
Quick Facts Names, Identifiers ...
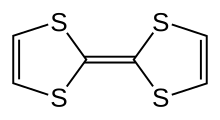 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2,2′-Bi(1,3-dithiolylidene) | |
| Other names
Δ2,2-Bi-1,3-dithiole | |
| Identifiers | |
3D model (JSmol) |
|
| 1282106 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.045.979 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H4S4 | |
| Molar mass | 204.34 g·mol−1 |
| Appearance | Yellow solid |
| Melting point | 116 to 119 °C (241 to 246 °F; 389 to 392 K) |
| Boiling point | Decomposes |
| Insoluble | |
| Solubility in organic solvents | Soluble[vague] |
| Structure | |
| 0 D | |
| Hazards[1] | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
combustible |
| GHS labelling: | |
 | |
| Warning | |
| H317 | |
| P261, P280, P302+P352, P333+P313, P363, P501 | |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close