
Fluorenol
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Fluorenol?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Fluorenol, also known as hydrafinil,[3] is an alcohol derivative of fluorene. In the most significant isomer, fluoren-9-ol or 9-hydroxyfluorene, the hydroxy group is located on the bridging carbon between the two benzene rings. Hydroxyfluorene can be converted to fluorenone by oxidation. It is a white-cream colored solid at room temperature.
Quick Facts Names, Identifiers ...
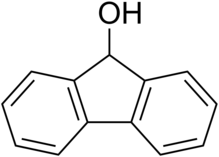 | |
 | |
| Names | |
|---|---|
| IUPAC name
9H-Fluoren-9-ol | |
| Other names
9-Hydroxyfluorene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.015.345 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H10O | |
| Molar mass | 182.22 g/mol |
| Appearance | Off-white crystalline powder |
| Density | 1.151 g/mL |
| Melting point | 152 to 155 °C (306 to 311 °F; 425 to 428 K) |
| Practically insoluble [2] | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
