
Hydroxyprogesterone caproate
Medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Hydroxyprogesterone caproate?
Summarize this article for a 10 year old
Hydroxyprogesterone caproate, sold under the brand names Proluton and Makena among others, is a medication used to reduce the risk of preterm birth in women pregnant with one baby who have a history of spontaneous preterm birth.[10] In March 2023, the manufacturer, Covis Pharma, agreed to withdraw the drug from the US market.[11][12][13] The approvals of Makena and its generics were withdrawn by the US Food and Drug Administration (FDA) in April 2023.[10]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Delalutin, Proluton, Makena, others |
| Other names | OHPC; Hydroxyprogesterone capronate; Hydroxyprogesterone hexanoate; 17α-Hydroxyprogesterone caproate; 17α-OHPC; 17-Hydroxyprogesterone caproate; 17-OHPC; 17-HPC; 17α-HPC; HPC; LPCN-1107; 17α-Hydroxypregn-4-ene-3,20-dione 17α-hexanoate |
| Routes of administration | Intramuscular injection,[1] subcutaneous autoinjection[2][3] |
| Drug class | Progestogen; Progestin; Progestogen ester; Antigonadotropin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: Very low (~3% in rats)[4] Intramuscular: 100% (in rats)[4] |
| Protein binding | Extensive (to albumin, not to CBGTooltip corticosteroid-binding globulin or (likely) SHBGTooltip sex hormone-binding globulin)[1][5][6] |
| Metabolism | Reduction and hydroxylation (via CYP3A4, CYP3A5, CYP3A7) and conjugation (glucuronidation, sulfation, acetylation)[1] |
| Elimination half-life | Non-pregnant: 7.8 days[7][8] Singlet: 16–17 days[1][9] Twins: 10 days[9] |
| Excretion | Feces: 50%[1] Urine: 30%[1] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.127 |
| Chemical and physical data | |
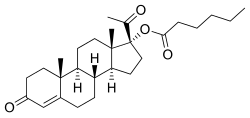
| Formula | C27H40O4 |
| Molar mass | 428.613 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Hydroxyprogesterone caproate is a progestin medication which was used to prevent preterm birth in pregnant women with a history of the condition and to treat gynecological disorders.[1][8][9][14][3] It has also been formulated in combination with estrogens for various indications (brand names Gravibinon and Primosiston) and as a form of long-lasting injectable birth control (brand name Chinese Injectable No. 1).[15] It is not used by mouth and is instead given by injection into muscle or fat, typically once per week to once per month depending on the indication.[1][4][3]
Hydroxyprogesterone caproate is generally well tolerated and produces few side effects.[1] Injection site reactions such as pain and swelling are the most common side effect of hydroxyprogesterone caproate.[1] The medication may increase the risk of gestational diabetes when used in pregnant women.[1][16] Hydroxyprogesterone caproate is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[16] It has some antimineralocorticoid activity and no other important hormonal activity.[17][18][19][20][21] The medication shows a number of differences from natural progesterone.[16][22]
Hydroxyprogesterone caproate was discovered in 1953 and was introduced for medical use in 1954 or 1955.[23] It was marketed in the United States under the brand name Delalutin and throughout Europe under the brand name Proluton.[24] The medication was discontinued in the United States in 1999.[25] However, hydroxyprogesterone caproate was subsequently reintroduced in the United States under the brand name Makena for the treatment of preterm birth in 2011.[26]