
Iridium(III) chloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Iridium trichloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Iridium(III) chloride is the inorganic compound with the formula IrCl3. The anhydrous compound is relatively rare, but the related hydrate is much more commonly encountered. The anhydrous salt has two polymorphs, α and β, which are brown and red colored respectively. More commonly encountered is the hygroscopic dark green trihydrate IrCl3(H2O)3 which is a common starting point for iridium chemistry.[4]
Quick Facts Names, Identifiers ...
 α-IrCl3 | |
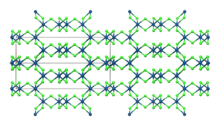 β-IrCl3 | |
 Iridium(III) chloride trihydrate | |
| Names | |
|---|---|
| Other names
Iridium trichloride | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.030.028 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| IrCl3 | |
| Molar mass | 298.58 g/mol (anhydrous) |
| Appearance | brown solid (α-anhydrous) red solid (β-anhydrous) dark green solid (trihydrate) |
| Density | 5.30 g/cm3, solid[1] |
| Melting point | 763 °C (1,405 °F; 1,036 K)[1][2] (decomposes) |
| insoluble (anhydrous IrCl3), soluble (hydrated derivative)[1] | |
| Solubility | Insoluble in HCl and alkanes[1] |
| −14.4·10−6 cm3/mol | |
| Structure | |
| Monoclinic, mS16 | |
| C12/m1, No. 12 | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
-257 kJ/mol |
| Hazards | |
| GHS labelling:[3] | |
  | |
| Warning | |
| H302, H411 | |
| Flash point | non-flammable |
| Related compounds | |
Other cations |
Rhodium(III) chloride |
Related compounds |
Platinum(II) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close