Loading AI tools
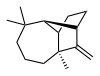
Longifolene is the common (or trivial) chemical name of a naturally occurring, oily liquid hydrocarbon found primarily in the high-boiling fraction of certain pine resins. The name is derived from that of a pine species from which the compound was isolated,[1] Chemically, longifolene is a tricyclic sesquiterpene. This molecule is chiral, and the enantiomer commonly found in pines and other higher plants exhibits a positive optical rotation of +42.73°. The other enantiomer (optical rotation −42.73°) is found in small amounts in certain fungi and liverworts.
 | |
| Names | |
|---|---|
| IUPAC name
(1R,2S,7S,9S)- 3,3,7-trimethyl- 8-methylenetricyclo- [5.4.0.02,9]undecane | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 5731712 2044263 4663756 | |
| ChEBI |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.812 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H24 | |
| Molar mass | 204.36 g/mol |
| Density | 0.928 g/cm3 |
| Boiling point | 254 °C (489 °F; 527 K) (706 mm Hg) |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H304, H317, H410 | |
| P261, P272, P273, P280, P301+P310, P302+P352, P321, P331, P333+P313, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Longifolene is also one of two most abundant aroma constituents of lapsang souchong tea, because the tea is smoked over pinewood fires.[2]
Terpentine obtained from Pinus longifolia (obsolete name for Pinus roxburghii Sarg.) contains as much as 20% of longifolene.[3]
The laboratory synthesis of longifolene has attracted much syntheses.[4][5][6][7][8][9][10]
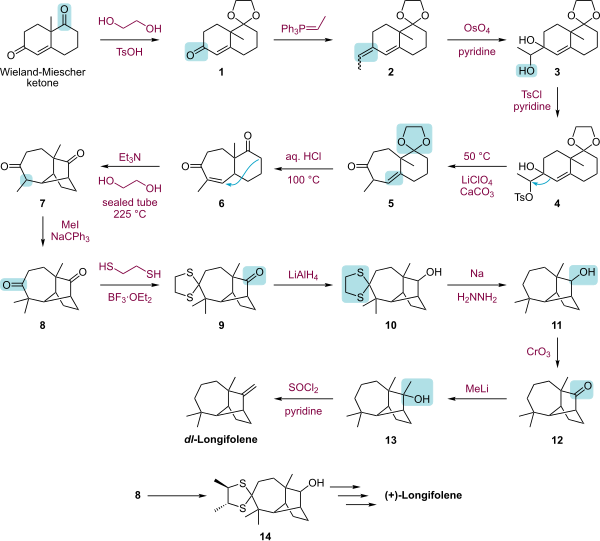 |
| Longifolene total synthesis by Corey.svg |
|---|
The biosynthesis of longifolene begins with farnesyl diphosphate (1) (also called farnesyl pyrophosphate) by means of a cationic polycyclization cascade. Loss of the pyrophosphate group and cyclization by the distal alkene gives intermediate 3, which by means of a 1,3-hydride shift gives intermediate 4. After two additional cyclizations, intermediate 6 produces longifolene by a 1,2-alkyl migration.

It reacts with borane to give the derivative dilongifolylborane, which is a chiral hydroborating agent.[11]
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.