Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
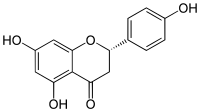
Naringenin is a flavanone from the flavonoid group of polyphenols.[2] It is commonly found in citrus fruits, especially as the predominant flavonone in grapefruit.[2]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
(2S)-4′,5,7-Trihydroxyflavan-4-one | |
| Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(4-hydroxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Naringetol; Salipurol; Salipurpol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.006.865 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.256 g·mol−1 |
| Melting point | 251 °C (484 °F; 524 K)[1] |
| 475 mg/L[citation needed] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The fate and biological functions of naringenin in vivo are unknown, remaining under preliminary research, as of 2024.[2] High consumption of dietary naringenin is generally regarded as safe, mainly due to its low bioavailability.[2] Taking dietary supplements or consuming grapefruit excessively may impair the action of anticoagulants and increase the toxicity of various prescription drugs.[2]
Similar to furanocoumarins present in citrus fruits, naringenin may evoke CYP3A4 suppression in the liver and intestines, possibly resulting in adverse interactions with common medications.[2][3][4][5]
Naringenin has the skeleton structure of a flavanone with three hydroxy groups at the 4′, 5, and 7 carbons.[2] It may be found both in the aglycol form, naringenin, or in its glycosidic form, naringin, which has the addition of the disaccharide neohesperidose attached via a glycosidic linkage at carbon 7.
Like the majority of flavanones, naringenin has a single chiral center at carbon 2, although the optical purity is variable.[6][7] Racemization of (S)-(−)-naringenin has been shown to occur fairly quickly.[8]
Naringenin and its glycoside has been found in a variety of herbs and fruits, including grapefruit, oranges, and lemons,[2] sour orange,[9] sour cherries,[10] tomatoes,[11] cocoa,[12] Greek oregano,[13] water mint,[14] as well as in beans.[15] Ratios of naringenin to naringin vary among sources,[2] as do enantiomeric ratios.[7]
The naringenin-7-glucoside form seems less bioavailable than the aglycol form.[16]
Grapefruit juice can provide much higher plasma concentrations of naringenin than orange juice.[17]
Naringenin can be absorbed from cooked tomato paste. There are 3.8 mg of naringenin in 150 grams of tomato paste.[18]
Naringenin can be produced from naringin by the hydrolytic action of the liver enzyme naringinase.[2] Naringenin is derived from malonyl-CoA and 4-coumaroyl-CoA.[2] The latter is derived from phenylalanine. The resulting tetraketide is acted on by chalcone synthase to give the chalcone that then undergoes ring-closure to naringenin.[19]
The enzyme naringenin 8-dimethylallyltransferase uses dimethylallyl diphosphate and (−)-(2S)-naringenin to produce diphosphate and 8-prenylnaringenin. Cunninghamella elegans, a fungal model organism of the mammalian metabolism, can be used to study the naringenin sulfation.[20]
The fate and biological roles of naringenin are difficult to study because naringenin is rapidly metabolized in the intestine and liver, and its metabolites are destined for excretion.[2][21] The biological activities of naringenin metabolites are unknown, and likely to be different in structure and function from those of the parent compound.[2][21]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.