Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
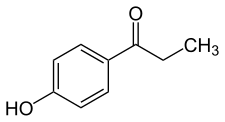
Paroxypropione, also known as paraoxypropiophenone, is a synthetic nonsteroidal estrogen which has been used medically as an antigonadotropin in Spain and Italy but appears to no longer be marketed.[1][2][3][4] It was first synthesized in 1902.[1] The antigonadotropic properties of the drug were discovered in 1951[3] and it entered clinical use shortly thereafter.[5]
 | |
| Clinical data | |
|---|---|
| Trade names | Frenantol, Frenormon, Hypophenon, Paroxon, Possipione, Profenone, others |
| Other names | Paraoxypropiophenone; H-365; NSC-2834; 4'-Hydroxypropiophenone; Ethyl p-hydroxyphenyl ketone; p-Propionylphenol; Paroxypropiophenone; Parahydroxypropiophenone; PHP |
| Drug class | Nonsteroidal estrogen; Antigonadotropin |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.676 |
| Chemical and physical data | |
| Formula | C9H10O2 |
| Molar mass | 150.177 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Paroxypropione is closely related structurally to p-hydroxybenzoic acid and parabens such as methylparaben, and also bears a close resemblance to diethylstilbestrol (which, in fact, produces paroxypropione as an active metabolite)[6][7] and alkylphenols like nonylphenol, all of which are also estrogens.[8][9] The drug possesses relatively low affinity for the estrogen receptor[4] and must be given at high dosages to achieve significant estrogenic and antigonadotropic effects, for instance, 0.8 to 1.6 g/day.[10][11] It possesses 0.1% of the estrogenic activity and less than 0.5% of the antigonadotropic potency of estrone.[12]
The highest reported yield, approximately 96%, is from the between phenol and propionyl chloride.[13] The mechanism is likely to involve initial esterification to give phenyl propionate, which then undergoes a Fries rearrangement.
Paroxypropione is a precursor in the chemical synthesis of diethylstilbestrol and dienestrol.[14][15]
Brand names Frenantol, Frenormon, Hypophenon, Paroxon, Possipione, Profenone, numerous others; former developmental code name NSC-2834, also known as paroxypropiophenone (P.O.P.) or 4'-hydroxypropiophenone.
Paroxypropione was studied and used in the treatment of breast cancer.[16][17][18]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.