Phosphite anion
Ion / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Phosphite anion?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
For phosphites of the type P(OR)3, see Phosphite ester.
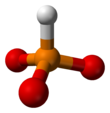
A phosphite anion or phosphite in inorganic chemistry usually refers to [HPO3]2− but includes [H2PO3]− ([HPO2(OH)]−). These anions are the conjugate bases of phosphorous acid (H3PO3). The corresponding salts, e.g. sodium phosphite (Na2HPO3) are reducing in character.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Phosphonate | |||
| Systematic IUPAC name
Phosphite[1] | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| 68617 | |||
| MeSH | Phosphorite | ||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| HPO2− 3 | |||
| Molar mass | 79.9810 g mol−1 | ||
| Related compounds | |||
Other anions |
Phosphinite | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close