
Reference electrode
Electrode with a stable and accurate electrode potential / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Reference electrode?
Summarize this article for a 10 year old
A reference electrode is an electrode that has a stable and well-known electrode potential. The overall chemical reaction taking place in a cell is made up of two independent half-reactions, which describe chemical changes at the two electrodes. To focus on the reaction at the working electrode, the reference electrode is standardized with constant (buffered or saturated) concentrations of each participant of the redox reaction.[1]
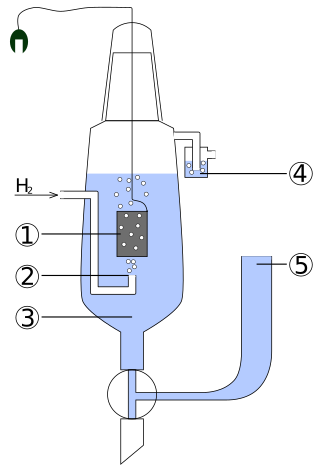
1) Platinized platinum electrode,
2) Hydrogen gas,
3) Acid solution with an activity of H+ = 1 mol/L,
4) Hydroseal for prevention of oxygen interference,
5) Reservoir via which the second half-element of the galvanic cell should be attached. The connection can be direct, through a narrow tube to reduce mixing, or through a salt bridge, depending on the other electrode and solution. This creates an ionically conductive path to the working electrode of interest.
There are many ways reference electrodes are used. The simplest is when the reference electrode is used as a half-cell to build an electrochemical cell. This allows the potential of the other half cell to be determined. An accurate and practical method to measure an electrode's potential in isolation (absolute electrode potential) has yet to be developed.