Loading AI tools
| This It is of interest to the following WikiProjects: | |||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
The first paragraph says:
- "...a valuable Lewis acid..."
does "valuable" have a specific chemical meaning, or does this just mean that it's costly to produce or extract from natural sources and therefore "valuable" in a monetary sense. Not really critical to the meaning of the article, I'm just curious. Thx. Zero sharp 19:32, 5 January 2007 (UTC)
"Valuable" here means that BF3 is a USEFUL Lewis acid. It is used in myriad applications, from catalyzing aldol reactions to removal of a variety of protecting groups. This is a common term in organic chemistry, though as you point out it can be misleading. Shultzc (talk) 03:13, 25 March 2008 (UTC)
My chemistry book explicitly says this: File:BF3 illustration.png
I'm a bit confused on this. Juraj5 (talk) 10:44, 3 May 2008 (UTC)
- This is straightforward. BF3 is planar, not pyramidal. Unlike NH3, BF3 only has six valence electrons around its central atom, all of which are in B-F bonds. There is no lone pair on boron, whereas nitrogen in ammonia has a lone pair, which repels the three N-H bonds, resulting in a pyramidal molecule.
- Thank you on your clarification! In this case, why is the article page showing BF3 having pyramidal structure? Juraj5 (talk) 18:30, 5 May 2008 (UTC)
- It doesn't. The image in the article shows a planar molecule, with on B-F bond in the plane, one coming out towards you and one going inwards away from you - but all in the same plane. This 2D image exactly follows the 3D image to its right. By contrast, the images at ammonia show a pyramidal molecule.
 |  |
 |  |
 |  |
- The fact that you're confused goes to show that structural formulae are confusing to the uninitiated. That's why I spend most of my time adding 3D images of molecules, because they're a lot easier to decipher, and people with little formal training in chemistry can easily understand what they mean.
Inorg. Chem., 1997, 36 (14), 3022–3030 suggests that:
- There is very little F→B π back bonding in BF3 (Greenwood and Earnshaw acknowledge that a small amount of F→B π charge transfer would lead to the same bond energy contribution as a much large amount of I→B π charge transfer - p. 196)
- BF3 has a lower Lewis acidity than BCl3 and BBr3, not because of better F→B π back bonding, but because upon forming a tetrahedral Lewis adduct, the B-X σ bonds lengthen and weaken, and this is more destabilising with B-F bonds (which are stronger than B-Cl bonds and thus have more stabilisation to be lost upon lengthening).
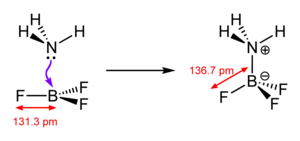 |
 |
This is alluded to in the article at present, but is presented as an alternative explanation. It seems, though, that the above is a better explanation, compatible with calculated partial charges (B: +2.43, F: −0.81) which indicate BF3 is predominantly an ionic molecule. The fact that BF3 does not condense to an ionic solid is explained by "size limitations on the maximum coordination number" that boron can achieve. If BF3 adopted the AlF3 structure (ionic solid), boron would be octahedrally coordinated by six fluorides.
Does anyone have any particular expertise in this area, or know of more recent conclusions?
Ben (talk) 14:41, 24 January 2009 (UTC)
- I came across the above as one of a series of papers by Gillespie et al (see LCP theory). I think that the various arguments all need to be described - there seems to be fair amount of educational capital invested in the π bonding story for example. Personally I am swayed by Gillespies arguments- but he is a good communicator. Looking at our BF3 article again, the different explanations could be much better spelt out. Give it a shot! --Axiosaurus (talk) 17:52, 24 January 2009 (UTC)
Okay, I'll put it on my list! Thanks for the reply.
Ben (talk) 19:14, 24 January 2009 (UTC)
- A user recently wanted to use BF3 as an example of backbonding on the π backbonding page. I found this ridiculous but apparently there are people using this language in the literature. I temporarily don't have journal access so I can't check for myself this information for myself. My question is why would someone call a π F→B electron transfer backbonding? I would call it a pi donation from a Lewis base to a Lewis acid while back bonding would be pi donation from "Lewis acid" to a "Lewis base" where the acid character is based on the sigma structure. I've always understood "backbonding" to mean a sigma acceptor donated its electron density "back" through the pi structure into the pi structure of the sigma donor. Even more specificity I thought it had to be a donation into a pi* orbital. What am I missing?--OMCV (talk) 05:05, 10 February 2010 (UTC)
- I looked at a few abstracts and I have an addition question. Ben when you said "F→B π back bonding in BF3" would you have also been able to say "O→C π back bonding in CO"? Is this really a discussion of whether the (debated) B-F pi bond is available to donate into the pi* of a Lewis base like CO?--OMCV (talk) 05:22, 10 February 2010 (UTC)
I was just using the terminology in the paper cited above. I can send you a copy if you like. I think your definition of π back bonding sounds like the most usual usage. Maybe F→B π donation is sometimes termed back bonding because it is in the opposite direction to the usual B-F bond polarity, with the much more electronegative fluorine drawing electron density away from boron. Ben (talk) 15:03, 10 February 2010 (UTC)
- I see what you mean but it still seems as if the authors are mixing models/metaphors. In the classic example of Fe-CO backbonding we would say there is Fe→C π donation, ie the less electronegative donates to the more electronegative, still the opposite of F→B π donation in terms of electronegativity. Thanks for offering to e-mail the paper but I will have journal access again in about a week.--OMCV (talk) 15:19, 11 February 2010 (UTC)
i find it very... irritating that in the equation boric acids formula is in parenthesis. either remove parenthesis or write as H3BO3
Steve Z: Previously listed anhydrous density of 2.167 g/cm3 was misleading. For liquid anydrous BF3, Carl Yaws' Chemical Properties Handbook liquid density curve ranges from 0.549 g/cm3 at critical temperature (-12.3 C) to 1.678 g/cm3 at -127 C, with 1.572 g/cm3 at atmospheric boiling point. Anhydrous BF3 can't be liquefied at 25 C, new density is ideal-gas density at 25 C and 100 kPa. —Preceding unsigned comment added by 216.203.59.252 (talk) 21:28, 16 April 2010 (UTC)
not sure where to put this:
Boron trifluoride was discovered in the autumn of 1808 by Joseph Louis Gay-Lussac and Louis Jacques Thénard, who were trying to isolate fluoric acid by combining calcium fluoride with vitrified boric acid; the resulting vapours failed to etch glass, so they named it fluoboric gas.<ref>“Sur l’acide fluorique,” Annales de chimie, 69 (1809), 204–220, written with Thénard; “Des propriétés de l’acide fluorique et surtout de son action sur le métal de la potasse,” in Mémories de physique et de chimie de la Société d’Arcueil, 2 (1809), 317–331, written with Thénard.</ref> -- 99.233.186.4 (talk) 01:35, 21 July 2010 (UTC)
I don't quite understand this section: Unlike the aluminium trihalides, the boron trihalides are all monomeric. They do undergo rapid reversible dimerization as indicated by the high rate of the halide exchange reactions:
- BF3 + BCl3 → BF2Cl + BCl2F
That reaction doesn't show a dimer. Is the intent of this to imply that this reaction must have an intermediate transient dimer of B2F3Cl3 ? If so, perhaps the intermediate step should be mentioned, and the word "transient" added before "dimerization."
Also, since the word "reversable" is used, this should show a two-directional arrow, not a one-directional arrow. Geoffrey.landis (talk) 14:29, 11 October 2011 (UTC)
Most chemists encounter BF3 as its etherate, but we dont have an article on that reagent. So I am planning to convert the redirect entitled boron trifluoride etherate. If folks see any problems with my plans, leave a note here or on my talk page.--Smokefoot (talk) 20:29, 11 August 2017 (UTC)
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.


