Loading AI tools
| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 1 |
It could be useful to mention that the proton is one of the very few "stable" particles around. Although the proton half life is given in a table, the layman may not realize that the proton and the electron are the definitive "stable particles" of elementary particle physics. —Preceding unsigned comment added by 62.12.66.243 (talk) 04:19, 15 June 2008 (UTC) Whats a proton? —Preceding unsigned comment added by 60.242.116.83 (talk) 12:55, 30 June 2009 (UTC)
While grammatically correct and well written, when viewed from the perspective that the goal of an encyclopedia is to impart knowledge to those who have none, this page is a dismal failure. As a layman, I can't understand any of this. A person would have to have a degree in physics or a related field to even understand this highly technical examination of protons- but if one already has that degree, what would they need this page for? Could some one take this piece of mental masturbation and add some explanation that would be of some use to the average reader? —Preceding unsigned comment added by 65.23.116.46 (talk) 10:49, 25 May 2008 (UTC)
I agree ! I have a PhD in engineering, and know something about the history of science, but I object strongly to 'his scintillatin detectors revealed the signiture of hydrogen atoms' (History section). What nonsense - what utter crap! Can hydrogen atoms write ? What does this mean ? Rutherford had some reason for identifying these particles as hydrogen nuclei, but this author clearly has no conception of what it was, yet it is a vital detail. This way of disguising ignorance with a meaningless metaphor just does not wash. —Preceding unsigned comment added by 82.32.49.157 (talk) 17:43, 7 January 2011 (UTC)
- Agreed here too. Articles of this nature should be written in a style and format that anyone can understand. In other words, plane english with necessary explainations. Sunshine Warrior04 (talk) 12:46, 4 February 2012 (UTC)
- Plane english is what we use to describe aircraft, not protons. <g>. "The power of instruction is seldom of much efficacy except in those happy dispositions where it is almost superfluous." -Gibbons, cited by Feynman in his preface to his "Lectures on Physics". Teaching to many audiences at once is therefore very difficult, and perhaps impossible. We can add some introductory material, but in the end, if you haven't the background, you're going to suffer. For example, if you look up the word "signature" you find it has many meanings (definition number 8, the figurative one, being the one in use here). SBHarris 19:08, 4 February 2012 (UTC)
Why doesn't it decay into an up quark? Quantum Burrito 22:50, 5 January 2007 (UTC)
- Don't be misled by the fact that the proton is lighter than the neutron - having less down quarks doesn't necessarily mean having less mass-energy. The lightest particle with 3 up quarks is the Δ++ which is more massive than a proton. I'm not sure of the reasons and I suspect they would involve some very complicated QCD. 80.7.20.133 18:47, 6 June 2007 (UTC)
It would be nice to have some information about the magnetic moment of the proton, (numerical value, theoretical understanding, etc.). The only reference to it I've seen in the article is in the section about the antiproton.
This is not a particularly interesting fact....
Interesting fact: the ratio of the mass of the proton to the mass of the electron is to 2 parts in 100000 equal to 6*pi^5.
I personally think that this is an interesting fact, of course I am almost finished with a physics degree.
No? Well, items relating proton mass to Britney Spears are pretty short on the ground. I guess the fact that the product of the electrical permitivity and magnetic permeability of free space were very close [within experimental precision equal to] to the reciprocal of the square of the speed of light is also exceedingly boring. Except that the notorious crank Maxwell based electromagnetic field theory from this observation. So if the almost non-mentioned fact that the mass of the proton to the electron is to a high degree of precision a simple multiple of a simple power of pi doesn't spark some degree of interest in you, then I guess you're just not cut out to be a physicist. :-)
- The age the Universe had when the song ATWA from System of a Down's Toxicity when it was released was 3*pi^30 times the duration of the song itself, so what?
Roadrunner is correct, and I'm afraid you, 81.86.154.69, are not. Playing with things like N*pi^M, or change pi for e, or whatever, you can get almost any number. This has nothing to do with Maxwell's observation for the "product of the ...", because indeed it is not true, Maxwell was not doing numerology (as you are doing), he explicitly calculated the speed of an electromagnetic wave (= 1/sqrt(mu * epsilon) ) and compared it with the speed of light.
Moreover, today we know that protons are made of quarks, and that their mass is not the sum of the masses of the three quarks, because the binding energy amounts for a lot more. So there is, no relationship foreseen whatsoever.
- What's more, 2002 CODATA's value for proton-electron mass ratio is 1836.15267261±0.00000085, and 6*pi^5 is 1836,1181087116, forty thousand times the uncertainity less than that.--Army1987 15:56, 19 July 2005 (UTC)
JBC
Hello.
I´m a young spanish student and i´d like to give you my congratulations for this wonderfull encyclopedia,i´m really surprised of its whole content. Before beginning i´d like to give you my sorries because my english is not so good as i´d like it to be,but i hope i will be able to explain my self. Anyway be sure that i understand whatever in this encyclopedia you´re talking about.
Cause my low knowledge about Quantum mechanics (in comparison whith you but not whith my friends) i imagine that my doubt will not supose you a great work.
I'm confused about what you explain in Proton page. Here i can read that "the charge of proton must be equal to the charge of electron", naturally, to get forcing equilibrium. Some lines down i can read that "the positron has the same magnitud charge as the electron but oposite in sign". My doubt stribes in the following question: if the positron is the antiparticle of the electron and they have the same but oposite charge, and the charge of proton must beequal to the charge of electron (and, naturally, basing my self in the fact that positron, charge +, electron, charge -; that means to me that charges are also oposite), here i have a contradiction for my mental schema. This make me think by any way that protons and positrons are the same particle, what i´m sure is not true.
So, concluding, my answer is: What kind of phenomenon i unknow makes different the behavior of proton and positron front the electron?
Thnks, :)
yikes, this whole article has no mention of the protons role in determining what element a substance is and what it does in chemistry. a proton is positive and an electron is negative and the neutron is negative. The atomic number is the number of protons in the atom and if you change the proton number you basically change the element. if you change the neutron number you can make it radioactive and if you change the election number you can change it electrically and chemically. 72.179.185.73 02:46, 26 April 2007 (UTC)
- The mass of a proton is much larger than that of a positron. - Patrick 20:04, 11 Dec 2003 (UTC)
- Unlike the positron, the proton feels the strong nuclear force. —User:Herbee 2004-02-10
This page should be a disambiguation page and the stuff should be moved to Proton (physics). Bensaccount 01:20, 28 Feb 2004 (UTC)
- I disagree. I mean, the physics related "Proton" is more likely to be the wanted subject of the artile over the "proton" title. So, i think there should be a direct redirection to the "proton (physics)" article, and a link to the disambiguation page. Kieff 04:59, Apr 11, 2004 (UTC)
You are right. My mistake. Bensaccount 17:32, 16 May 2004 (UTC)
I'm a little confused, why is mass measured in eV?
- It's a shorthand that physicists like to use. Particle physicist types are generally concerned with calculating and measuring energies rather than masses in particle reactions and such, and so they get to a rest mass by converting the rest energy with a factor of c2 (good ol' E=mc2). Being lazy, however, they tend to drop factors of c (and h, Planck's constant) when doing calculations, sticking them back in when it's necessary to actually get a number out.
- However, this article isn't written for physicists, so I'll add the c2... Carmelbuck 03:56, 12 Nov 2004 (UTC)
- It's not laziness. c = 1 is part of a perfectly acceptable and useful system of units. -- Xerxes 03:34, 2004 Nov 13 (UTC)
- I don't knof if Xerxes' quote was a joke, but c=1 does not mean that it isn't there. It is still correct to include it. Pdbailey 04:06, 23 Nov 2004 (UTC)
- It's not a joke; it's the absolute solid truth. Particle physicists use a system of units where hbar=c=1. This system of units is just as valid as any other system we choose, and in many ways can be considered more natural. This has some side effects that seem a bit odd to people who have only learned classical mechanics (energy and mass have the same units, for instance; and the units of mass are inverse the units of distance) but that's all they are---odd to people not used to them.
System of units with c=1 adimensional valid?Same units for mass and energy?
c=1 is just numerical value with what unit of measurement? c beeing a speed has dimension length/time, so c=1 must have units. If c=1 is adimensionalized other dependent quaantities must be adimensionalized.--84.232.141.38 (talk) 11:46, 9 May 2010 (UTC)
News
teleronki: Newly calculated elementary particle of rest mass = 1.15819171.10^-30 kg which in is included as integer value in the rest mass of proton and neutron. (Source: Meissner, R.:Das Teleronki-Modell..., Aachen: Shaker-Verlag 2001.)
I'm removing this:
"Because the electromagnetic force is many orders of magnitude stronger than the gravitational force, the charge on the proton must be equal and opposite to the charge on the electron. Otherwise, the net repulsion of having an excess of positive or negative charge would cause a noticeable expansion effect on the universe, and indeed any gravitationally aggregated matter (planets, stars, etc.)."
The argument that we must be living in an approximately charge-neutral observable universe is correct. The conclusion that protons and electrons have the same charge makes the assumption that they occur in equal numbers. I see no basis for such an assumption; they could have different charges and occur in correspondingly different numbers. This would not be a "coincidence" cosmologically if there were a good reason for charge neutrality; the particles we end up with would have been constrained by that.
There are good theoretical reasons to believe that the charges of protons and electrons are exactly equal; perhaps such belong on the page about electric charge.
- Somebody restored the above claim of evidence of charge equality to the page. It appears to be fallacious, and whoever restored it did not address the dispute on this talk page. So I'm taking it back out. Dhochron 22:01, 26 October 2005 (UTC)
Does anyone know if there are any theories to the possibility that the Down Quark is composed of an Up Quark and an electron? I once heard that when a Proton and Electron mix it produces a neutron. So if this is true then if you mix an Up Quark and an electron it must form a down quark...I am not sure though. - BlackWidower
- The best theories that we have allow particles to decay into other particles without being "made of" the other particles. One way of saying it is that the particle fields are coupled; when the quantized self-propagating mode of one field (e.g. a particle) decays, it excites modes in the other fields due to the coupling. However, the behavior of the particle before the decay does not act like it is "made of" the decay products. In fact, some particles have multiple decay modes, but before they decay they can be proven to be identical
A proton would repel an electron because of opposite charges. The down quark is an indivisible quanta of matter, therefore it isnt "composed" of anything, it is indivisible.
Eugene Goldstein discovered the proton. Bayerischermann 00:26, 26 October 2005 (UTC)
- Do you have evidence for this claim ? According to PBS, Encarta and ScienceWorld , Rutherford discovered the proton.
- This claim is completely correct . For evidence you can browse through Class 9th N.C.E.R.T Science book (INDIA). E . Goldstein during a gas discharge found positively charged radiations which he name canal rays. Also that E.Goldstein discovered protons in 1886 way before discovery of electrons (1900's) by J.J.Thompson. E.Goldstein also concluded that protons had a charge equal to electrons but opposite in sign (postitive) and that the mass of 1 proton is equal to 1 unit (like that of neutrons) and that of electtron is negligible.--Pivren (talk) 07
- 23, 18 May 2010 (UTC)
- ---
- The claim is incorrect. Canal rays are positive ions not protons or bare nuclei (except in the case of hydrogen). See, for example the presentation of the 1919 Nobel Prize in Physics to Stark:
- In the year 1886 Goldstein discovered a new kind of rays in discharge tubes containing rarefied gas, the study of which has become extremely important to our knowledge of the physical properties of atoms and molecules. In view of the manner of their formation Goldstein called them canal rays. It was proved by the research of W. Wien and J.J. Thomson that the majority of these are composed of positively charged atoms of the gas in the discharge tube, which move along the beam at a very high velocity. (my emphasis)
- As an entity distinct from ordinary ions, it would be incorrect to say that the observation of canal rays amounted to the discovery of protons. Rather, it was Rutherford who:
- 1) determined that the proton is small and dense
- 2) proposed that it makes up the nuclei of all atoms
- which is really what defines the proton.
- Anyway, the history paragraph could add a bit on Goldstein without explicitly claiming he "discovered" the proton. -- Xerxes 14:51, 26 October 2005 (UTC)
- It's not my claim, it's what both my Chemistry teacher and my textbook (Wilbrahm, Anton C, Dennis D Staley, and Michael S Matta 1995. Chemistry. Menlo Park, California: Addison-Wesley) say. In particular, it says "In 1886 E. Goldstein, using a cathode ray tube in which the cathode had holes, observed rays traveling in the opposite direction to the cathode ray. These rays, shown in figure 4.5, contain positively charged particles and are called canal rays." and to the left of Figure 4.5: "If the gas in the cathode ray tube was hydrogen, the canal rays would be made up of protons; after hydrogen gas atoms lose electrons at the cathode, only protons remain to form the canal ray." In the book, this is listed as the first discovery of protons. Rutherford was after Goldstein, and I never even suggested that Goldstein influenced anybody. Bayerischermann 21:49, 26 October 2005 (UTC)
- Xerxes summed it up nicely, i.e. observing canal rays is not equivalent to discovering protons, and something on Goldstein should be added. Since only one source seems to claim Goldstein discovered the proton, and the remaining ones claim Rutherford, I would suggest -- without further evidence -- that the one source was wrong Salsb 21:58, 26 October 2005 (UTC)
Reprise 2009
I'd like to reopen this argument. To say that Rutherford "discovered" the proton is misleading and ignores previous scientists who suggested its existence. It is true that from Goldstein's work, it emerged that atoms contain both positive and negative elements, a fact that was confirmed by Thomson's work. The negative part was directly interpreted as a new particle, the electron, from Thomson's work because it produced a single e/m measurement; whereas the positive part (canal rays) did not, and there was a long period (roughly 1897 - 1919) when its precise nature was unknown. Rutherford originally thought it was alpha radiation, ie that all nuclei are multiples of helium nuclei (except hydrogen?); but it became increasingly clear, first van den Broek 1911, then Moseley 1913, that the fundamental positive charge is the hydrogen nucleus. Rutherford`s 1919 "discovery" was only the final nail to the whole line-of-argument, so to speak. By Xerxes' criteria, Rutherford did NOT discover the proton, because he did NOT show the hydrogen nucleus to be small and dense (his experiments were with other nuclei), and he did NOT propose that atomic nuclei contained the hydrogen nucleus (others proposed it but he proved it). --85.232.196.151 (talk) 16:38, 28 August 2009 (UTC)
I have now rewritten the History section and tried to put Rutherford in his proper context. For one thing I have followed the historical order - Prout, Goldstein, van den Broek, Moseley, Rutherford. Dirac66 (talk) 14:48, 7 September 2009 (UTC)
I'm no expert, but one part in 10-8 seems like an awfully big uncertainty. Shouldn't this be one part in 108?
The following was apparently cut-and-pasted from an AIP news article. In its present form, it is certainly unsuitable for the article; perhaps it would be more suitable (heavily edited) in a HAPPEx article? In addition, the original content author demands a specific form of credit for reposting this content.
- ...
-- Xerxes 15:36, 2 May 2006 (UTC)
- I assume that was a copyright violation, so I've deleted it here too. Melchoir 16:17, 2 May 2006 (UTC)
This seems a bit silly, and I was a physics major, but... what's the diameter of the proton? We can all recite the characteristic size of the hydrogen atom (1 angstrom, 10^-8 cm, 10^-10 m), and it's said that the proton is not pointlike but has some diameter. So what is it? I found one source that suggested, without attribution, that it was on the order of 10^-15 m. Is this firm? Is it quantum-uncertain? Should it be part of the basic attributes like mass, charge, etc.? Google searches for "proton diameter" were not conclusive.Eh Nonymous 20:06, 23 May 2006 (UTC)
- It's a bit hard to say what "proton diameter" means. Most experiments seem focused on determining the "proton root-mean-squared charge radius". This value is not very well known, but is about 0.87 fm. So by that measure, the proton diameter is about a fermi and a half. -- Xerxes 21:07, 23 May 2006 (UTC)
The propton-diameter mentioned now under properties (1.5*10-15 m.) is half the electron radius mentioned in Mass_of_electron. Unfortunately I don't know what to trust, but probably something is not OK. (Jan-Willem, 21 July 2006)
- The "electron radius" mentioned there is the Classical electron radius - based on non-quantum physics and the assumption that the electron's mass is due to electrostatic potential energy, i.e. based on assumptions which are known to be flawed. Maybe that should be made clearer in the electron article. HairyDan 18:35, 6 June 2007 (UTC)
Charge radius is referenced above. There's no article on it and it isn't defined in the proton article. Somebody needs to do one or the other. 4.249.3.140 (talk) 20:20, 22 May 2009 (UTC)
 Done Charge radius. Physchim62 (talk) 23:07, 15 July 2010 (UTC)
Done Charge radius. Physchim62 (talk) 23:07, 15 July 2010 (UTC)
Will someone please explain charge radius?
It is now July 2010 and today's edits are concerned with whether the best value of the proton charge radius is 0.84184 fm or 0.8768 fm. But in the infobox ... the link to charge radius from the infobox is still RED, which on Wikipedia means that it is a dead-end which does not (yet) lead to an article. Would someone please start an article on Charge radius to explain the meaning of this term, as well as the mathematical definition and its justification (I think root-mean-square is involved, but why), and briefly how it is measured? I suggest a separate article rather than a section of this article, because I presume that the question applies to other particles as well as the proton. Dirac66 (talk) 00:25, 10 July 2010 (UTC)
- not competent to do this, but Charge density#Quantum charge density defines charge density for quantum objects in terms of their wave-function. So this looks like charge measurement as a distribution over a volume. If it were a classical function over a linear domain you'd calculate the Root mean square as the square-root of the mean of the squared values of the function over the domain. So it is some generalisation to a 3d domain (volume) of a (probability) distribution rather than a function. It looks like it is measured using quantum theory applied to the proton's wave-function, to relate to something physically observable about the proton. Puzl bustr (talk) 18:32, 12 July 2010 (UTC)
- Very briefly, because I'm not really competent to do this either, the "charge radius" is the apparent radius of the proton: I say apparent, because the proton does not have a firm boundary. You can measure the charge radius by electron–proton scattering (apparently, see Mohr, Peter J.; Taylor, Barry N. (1999). "CODATA recommended values of the fundamental physical constants: 1998" (PDF). Journal of Physical and Chemical Reference Data. 28 (6): 1713–1852. Bibcode:1999JPCRD..28.1713M. doi:10.1063/1.556049. Archived from the original (PDF) on 2017-10-01. at 1737); the proton radius also enters into the theoretical corrections that must be made to experimental transition frequencies to obtain the Rydberg constant (see Mohr, Peter J.; Taylor, Barry N.; Newell, David B. (2008). "CODATA Recommended Values of the Fundamental Physical Constants: 2006" (PDF). Reviews of Modern Physics. 80 (2): 633–730. arXiv:0801.0028. Bibcode:2008RvMP...80..633M. doi:10.1103/RevModPhys.80.633. Archived from the original (PDF) on 2017-10-01. at 16). The current CODATA recommended value is 0.8768(69) fm. Physchim62 (talk) 17:39, 13 July 2010 (UTC)
Measurement
Some details about the methods of measurement of charge radius should be included.--84.232.141.36 (talk) 13:15, 20 January 2011 (UTC)
- This can now be found at Charge radius#Modern measurements. Dirac66 (talk) 14:04, 20 January 2011 (UTC)
Measurement from ionic crystals diffraction
Are there available data of proton diameter measured from diffraction of ionic metal hydrides (like sodium hydride)? It would be interesting to compare these data with those from other methods.--86.125.162.153 (talk) 21:17, 21 February 2011 (UTC)
- Is there some ionic compound where it H positive ion could be found? —Preceding unsigned comment added by 86.125.162.153 (talk) 21:27, 21 February 2011 (UTC)
How about HCl crystals? Is the hydrogen ion present?--79.116.77.129 (talk) 22:31, 22 February 2011 (UTC)
- No. Pure solid and also pure liquid hydrogen chloride are in molecular form. HCl only becomes ionic when water is added. Dirac66 (talk) 03:29, 23 February 2011 (UTC)
Is it possible to determine the radius of a proton from diffraction measurement data of solid hydronium salts like hydronium perchlorate?--82.137.8.179 (talk) 15:53, 14 March 2012 (UTC)
- Do not confuse the chemist's hydrated proton with the physicist's bare proton. X-ray diffraction of H3O+ ClO4- can provide the O-H bond length in the H3O+ ion, from which we can subtract the approximate single-bond radius of oxygen to obtain a hydrogen radius of about 20-30 pm. But this refers to a proton with its share of the electron cloud forming the O-H bond. In contrast the physicist's proton is in vacuum with no electrons nearby, and its radius is less than 1 fm (104 times smaller) as explained in the section Charge radius. Dirac66 (talk) 02:48, 15 March 2012 (UTC)
These references at Further reading section were deleted as original research:
- The following analysis addresses the problem of proton creation and it's integrated in the concepts of the Physics of Creation:
- Aspden, Harold (2003), Physics of Creation: The Creation of the Proton (Chapter 4), PhD. Physics - University of Cambridge [1953], UK [pdf file]
- Aspden, Harold (2005) Aspden Research Papers Nº4: The Creation of the Proton
- "The value that they [Aspden and Eagles] calculate is remarkably close to our experimentally measured value (i.e. within two standard deviations)This is even more curious when one notes that they published this result several years before direct precision measurements of this ratio had begun." R. S. Van Dyck, Jr., F. L. Moore, D. L. Farnham and P. B. Schwinberg in Int. J. Mass Spectrometry and Ion Processes, 66, p. 327, 1985.
This author seems to have some peer-review published papers ('Physics Today', 'Journal of Applied Physics', 'American Journal of Physics', 'Physics Letters', 'Hadronic Journal', 'Physics Essays', Physics Education', etc.) and his work is mentioned by other members of the scientific community (as quoted above). And now his work cannot be presented in the related Wikipedia's articles??? What policy is this?! --88.214.171.194 05:26, 8 September 2006 (UTC)
- The paper you refer to has been superseded by a paper from the same authors, in which they report a better value of the ratio, which seems to rule out the value given by Aspden. (Phys. Rev. Lett., Vol.
7075, p. 3598) They also make no mention of Aspden in this newer paper. - Moreover, the particular writings you link to are clearly original research. (Self-published works constitute original research.) The fact that the author has been published in legitimate journals -- apparently on other topics -- does not mean that all of his publications are acceptable. MOBle 05:41, 8 September 2006 (UTC)
- It is on volume 75 , and the measured value "via Penning Trap Mass Spectroscopy" of 1836.152 666 5(40) (1995, 20 years later) is slightly above 1836.152, as it was recorded in Aspden's 1975 paper of 1836.15232 (H. Aspden & D. M. Eagles, Il Nuovo Cimento, 30A, 235 (1975)) , a theoretical value derived from his aether theory (not from wide expensive experiments). Still, how was he able to derive this value so close to the experimental result and not be considered fit as a resource to a Wikipedia article? Worse, discarding his work when you donnot have other theorie(s) than can predict such accuracy? Interesting that in 1960 and 1977 Dr. Aspden already published about the "electrostatic spin" (described as 'aether spin' induced by electrostatic charge ) before its discovery was announced by the University of California in April 2003 (note that this phenomenon could not be explained by available theory at the time of its discovery). Something strange is going on in the Physics field... and I am not even a physicist (people like me is just start noticing these unbelievable things). Really, I sense you should give a better look to his 2005 paper (above). Thank you for your attention. --88.214.171.194 06:29, 8 September 2006 (UTC)
- As you point out, Aspden's prediction was 1836.15232. This is 87 standard deviations away from the accepted value of 1836.1526665(40) cited above. That essentially means that Aspden's value is ruled out scientifically. That is the reason it should be discarded: experiment proved it wrong. His theories don't seem to explain anything.
- You may find his writings very interesting. You may even be convinced by his claims that he's predicted unusual physical effects before they were discovered by others. However, until his claims are verified by other experts and published in reputable sources, they should not be included in Wikipedia. MOBle 06:58, 8 September 2006 (UTC)
- Neither the mentioned experiments were conceived to test his theories (or the whole work), neither the result presented in his 1975 paper (and you have no other) was calculated in order to reflect the type of high precision measurement of such experiments (therefore he presents approximate values, yet accurate as proven by the 1985 measurement). Nevertheless, even the value you state as an "accepted value" seems to be different from the value given by CODATA (Rev. of Mod. Phys., vol 77, Jan 2005), and I think they are not the only values which were given to the measurement of the proton-electron mass-ratio through the last two decades. Last, I have already understood that the verification you mention to his "claims" will not be conducted, at least not by the current "experts", as in the aether vs. Relativity subtle war... If this was to be the case, it would have been conducted-done more than 10 years ago at least (since his work has already more that 40 years of research and the Van Dyck's and fellows measurement confirmation occured about 21 years ago). Perhaps in a next generation of Physicists! :) --88.214.171.194 09:12, 8 September 2006 (UTC)
Is it possible to make the definition simpler to read.......Thank You —The preceding unsigned comment was added by 129.44.215.148 (talk • contribs) 22:14, 10 September 2006 (UTC)
What could be simpler than that? This isn't NeanderthalPedia, we expect people who are reading this article to actually be able to read it. But if it's necessary: A proton is a subatomic particle present in the nucleus of atoms (DO NOT ADD THAT TO THE ARTICLE). Slartibartfast1992 00:56, 12 April 2007 (UTC)
Not necessarily, as a proton is equivalent to an atom of hydrogen. Therefore, you must say it is a particle composed of two up quarks and one down quark. —Preceding unsigned comment added by 96.249.19.77 (talk) 01:47, 19 April 2010 (UTC)
- The proton has a density of about 2.31 × 1017 kg m−3.
That would imply a proton radius of 1.2 fermi—not one of the values mentioned here. Three significant digits also seems a bit too much. Probably somebody playing with their calculator. Since density is normally not a hot topic with particles, I removed the sentence.
—Herbee 20:35, 4 December 2006 (UTC)
- I think the density might be a useful fact for general readers, because it helps put into perspective how "empty" ordinary matter is. Itub 20:54, 7 December 2006 (UTC)
- Such a perspective might be useful, but the proton article wouldn't be an obvious place to look for information on the emptiness of ordinary matter. The Orders of magnitude (density) article already contains an entry for the proton.
—Herbee 00:25, 8 December 2006 (UTC)
- Such a perspective might be useful, but the proton article wouldn't be an obvious place to look for information on the emptiness of ordinary matter. The Orders of magnitude (density) article already contains an entry for the proton.
Why the rest mass of a proton is bigger than the mass of 3 free quarks? --Daniel bg 15:35, 13 April 2007 (UTC)
- Recalling mass-energy equivalence, the binding energy, which is the amount needed to overcome the strong interaction to free the quarks, I believe. — Rebelguys2 talk 16:54, 13 April 2007 (UTC)
- With any other force we would expect the mass to be less than that of the free particles (the difference being the binding energy, i.e. the amount of energy you have to add to get the particles free), but what we mean by "free" is a little different with quarks - see Asymptotic freedom. HairyDan 18:27, 6 June 2007 (UTC)
- No, it's very simple: the 3 quarks have a total rest mass of about 15 MeV. But they have moving so fast that their kinetic energy contributes the other 923 MeV to the rest mass (invariant mass) of this system, the proton. All the strong force does is keep them bound, even with that tremendous energy of motion. The proton is like a a massless bottle of hot gas, which is so hot that most of its mass is due to the kinetic energy of the gas, not the rest mass of the gas molecules (which is what what the bottle would weigh, if the gas were cold). SBHarris 23:46, 13 October 2008 (UTC)
Where does the lead sentence that says the mass is predictable come from? As far as I can see the Physical Review is full of articles today still trying to do this. Here's an example The Constituent Quark Model Revisited - Quark Masses, New Predictions for Hadron Masses Brews ohare (talk) 18:55, 2 December 2008 (UTC)
- Good question. I have now removed this word as requiring explanation. Dirac66 (talk) 20:04, 2 December 2008 (UTC)
--Can someone please explain to me How if scientists don't know the mass of an individual quark how can anyone say they know the energy each individual component of the system has??; E=mc^2??? If quarks can't exist as lone particles by definition, how in the world will you ever know what energy each quark possess individually?? What I mean is, Lets say I have some unknown amount of apples, all of the apples must fill up a barrel. Without observing the individual apple and measuring it's volume, how can I be certain 5 apples fill the container? might as well be 6 or 7 or 1000.. I see the same thing with Quarks, Scientists say there are 3 quarks to a proton, yet they can't observe a quark individually; Doesn't make a lot of sense, without being able to observe the individuals how can anyone say how much mass or how much energy a quark has or doesn't have??
If Quarks can't obey gravity because the math doesn't work, Why then do Protons and Neutrons obey gravity??? It's logically inconsistent. If electrons/protons obey general relativity (or even more consistently obey classical mechanics) by definition, Why then do it's component pieces not?? That's also logically inconsistent .. 1 apple + 1 pear a banana don't make..
I think before anyone jumps to the conclusion that quarks exist they need to fix the inconsistencies in their own theories, ----I can't very well go around saying magical toasters sooo small they can't be observed by any known method, and can't exist individually the sum of the magical toasters make up the fundamental particles of our world, and Oh btw the magical toasters don't obey the laws of our Universe yet they create our universe ; and be able to get away with my outrageous claim.. —Preceding unsigned comment added by 97.77.60.254 (talk) 18:34, 6 October 2010 (UTC)
- The formula refers to the fact that the mass-energy equivalence of a particle is equal to its kinetic energy as v approaches the speed of light. The formula for kinetic energy is:
- The problem is, an object's mass approaches infinity as well, so we have to disregard that. Substituting v for c, the speed of light, we obtain the following equation:
- I think the properties of quarks are predicted by quantum theory. And besides, you can observe quarks with a particle accelerator.
- By the way, there are 597863792824204508315503453.98052 protons in a kilogram. This should be the new definition of the kilogram! 68.173.113.106 (talk) 02:11, 7 December 2011 (UTC)
I have removed the ludicrous claim that the Beatles' "I'll be back" is about Eugene Goldstein's work on "canal rays" (just look at the lyrics) - this seems to have been added by a serial vandal at 161.53.73.35. HairyDan 18:21, 6 June 2007 (UTC)
I edited some obscene language, but it is missing some of the original language in the first part after the introduction. —Preceding unsigned comment added by 71.103.43.104 (talk) 23:17, 7 October 2008 (UTC)
Do you think it's possible I can create a "topped" proton, by taking a proton and shooting a super-high-energy W plus boson at the down quark and turn it into a top quark, thus making a baryon with quark composition uut, electric charge +2, and spin 3/2?--Mathexpressions 02:36, 12 July 2007 (UTC)
- There is any particle of electric charge +2. Only "He", but that is an helium atom and it has, at least, 1 neutron. I though such baryion is impossible. Althoug this superproton will decay, inmediatelly, into two protons, to preserve electrical charge, and, might be, other particles.—Preceding unsigned comment added by Coronellian (talk • contribs) 22:21, 20 August 2007 (UTC)
No, it is impossible because the W+ boson doesn't interact via the strong-force, only the weak force. Therefore, there would be no interaction with the proton and boson. Also, the W+ is a boson, which means the spins are different and the W+ does not obey the pauli exclusion principle. Therefore, the W+ would pass through the proton with no interaction. —Preceding unsigned comment added by 96.228.119.103 (talk) 03:23, 18 April 2010 (UTC)
- No, it's not possible, but all the reasons given above are completely wrong. The real reason it is not possible is that the top lifetime is to short for it to take part into any bound state. Dauto (talk) 22:24, 8 June 2010 (UTC)
- The anonymous who said there is no particle with electric charge +2 is also wrong. There is the Δ++ which has charge +2 and spin 3/2 as requested, but with quark composition uuu, not uut. SpinningSpark 08:37, 9 June 2010 (UTC)
- As Dauto and Spinningspark said, the early replies are wrong. Several ++ baryons exists, such as the
Δ++
(uuu) and the
Σ++
c (uuc) (see List of baryons for a full list). In theory, your "topped proton" (uut) would be a
Σ++
t (spin 1/2) (the spin 3/2
Σ∗++
t would be the equivalent of a "topped delta"). However due to the top quark's extremely short lifetime, these can never be observed. However, the charm quark is longer lived, so process of smashing a
p+
/
Δ+
(uud) and a
W+
can (and does) generate uuc baryons (
Σ++
c/
Σ∗++
c).Headbomb {talk / contribs / physics / books} 10:16, 9 June 2010 (UTC)
- As Dauto and Spinningspark said, the early replies are wrong. Several ++ baryons exists, such as the
can protons move i think only electrons can move to protons but yea —Preceding unsigned comment added by 60.234.233.76 (talk) 06:13, 16 November 2007 (UTC)
Protons move all the time, in fact. Accelerated protons constantly bombard our atmosphere and create second and third generation particles. Besides, all motion is relative. —Preceding unsigned comment added by 96.249.19.77 (talk) 01:49, 19 April 2010 (UTC)
Part —Preceding unsigned comment added by 70.121.250.204 (talk) 23:11, 13 October 2008 (UTC)
I updated the physical properties of the proton using C. Amsler et al., "Review of Particle Physics" Physics Letters B667, 1 (2008). However, I am new at this and do not know how to put in multiple citations to this without creating several entries in the reference list. Someone who knows how should probably insert these citations on the appropriate places (proton mass, proton - or rather elementary - charge, etc.), at least if we want references in the info-box. I took the proton lifetime from the original reference (citet in the article). --Blennow (talk) 22:38, 20 October 2008 (UTC)
Why are there two very different values for the experimental lower bound on the lifetime? First 1035 years without a reference, and then two lines further (and also in the infobox) 2.1 x 1029 years properly referenced. Does this mean that a) the first value comes from a more sensitive experiment and just needs a reference, or b) the first value is wrong and should be deleted? Dirac66 (talk) 20:15, 2 December 2008 (UTC)
- I believe that the 2.1 x 1029 year is the lower bound on the p → invisible decay (i.e., proton to anything), while the 1035 year value refers to decays with visible decay products. Well, actually, looking at the latest version of the PDG, the lower bound on visible decays is 1031 or 1033 years depending on assumptions, so the larger value is a mystery to me. --Blennow (talk) 22:40, 2 December 2008 (UTC)
- Thank you. There are two different experiments then. As a chemist, I would like to better understand what is looked for in each experiment, without reading papers in particle physics.
Question 1. The proton decay article says that the usually expected products are positrons and neutral pions (which decay into two gamma), so does the visible decay value refer to a search for positrons and/or neutral pions and/or gammas?
Q2. Does the lower bound on proton to anything imply some direct measurement of the number of protons remaining, or perhaps their total mass? If so, is this intrinsically less sensitive than detection of visible products? That would explain the difference in the two bounds on lifetime without having to postulate more exotic products. Dirac66 (talk) 03:13, 9 December 2008 (UTC)
Is there any special reason why we have such a short and rather unreferenced article about the proton, compared to Neutron and Electron? Did just nobody care to expand it? --Apoc2400 (talk) 21:51, 2 February 2009 (UTC)
Hmm. Proton 10K bytes, neutron 24K, electron 103K. But we don't expand an article just because others are longer. What should be added to the proton article? Dirac66 (talk) 02:14, 3 February 2009 (UTC)
I agree this article could be expanded and better referenced. What exactly could be added should be debated, but talking more about its role in atomic nuclei and in how many of them there are in a given nuclei determines what chemical element it will form when combined with electrons would certainly be a start, being the dominant chunk of the "chemistry" section, rather than something about acids (not that this should be removed). The history of protons should probably contain more about their discovery, possibly a diagram/schematic of the Geiger-Marsden experiment, and mention the Rutherford model. A discussion about other atomic models / nuclear models would be in order too. The role of the proton related to the other baryons (not only the neutron) could also be mentioned, especially Delta baryons, possibly leading into a discussion of the Eightfold way.Headbomb {ταλκκοντριβς – WP Physics} 03:09, 3 February 2009 (UTC)
This seems a good list to start. I have now taken the first step by revising the chemistry section as suggested. Dirac66 (talk) 03:22, 4 February 2009 (UTC)
Exposure during flight... here is another study. http://www.sciencemag.org/cgi/content/abstract/216/4549/939
- I was curious if the Columbia Space Shuttle Flight was radioactive. (2 Feb 2003). I remember health warning about this, such as this article, but there is no corelation made with protons. --CyclePat (talk) 16:20, 21 September 2009 (UTC)
This may be an obviously newbie question, for which I apologize. Many of the values given in the sidebar have a number in parentheses following the fraction; for example, the mass of the proton is given as "1.672621637(83)×10−27 kg". I've never seen the "(83)" notation before, wonder what it means, and suggest that an explanation be given somewhere. I've looked at the "Notation" article and followed several leads in the "Physics" area, but so far have come up empty. About all I can guess is that the value is known to 83 digits, or there are 83 additional digits known, or something like that. SpiffZarf (talk) 12:04, 18 May 2010 (UTC)
The 83 is the estimated measurement uncertainty in the last digits. 1.672621637(83) is the concise notation for 1.672 621 637 ± 0.000 000 083. See Uncertainty#Measurements. Dirac66 (talk) 15:02, 18 May 2010 (UTC)
I have yet to read an article which makes a claim about the fundamental particles which actually explains the science behind their claims, Gluons have never been observed to exist yet the article presents an opinion that yes indeed Gluons exist and they hold the proton together.. -- Until there is science to prove the hypothetical Gluon Wikipedia should delete this section from the article since it's only conjecture and not really science.
Also on the topic of the quark building up the proton/neutron and other fundamental particles -- It's my observation that there is little conversation on how scientists KNOW Quarks exist; There are clear inconsistencies in the explanations of how a scientist knows that something exists when they have no proof of it's existence in a laboratory environment ie it's NEVER been observed, especially when there are other theories which don't require these fundamental particles.
To begin, both neutrinos and quarks cannot be directly detected as physical particles in
our spacetime. Consequently, all claims on their existence are indirect, that is, based on the
detection of actual physical particles predicted by the indicated theories
So Someone please edit the Quark section so that it is more moderate and does not say things which cannot be confirmed scientifically..
http://www.i-b-r.org/Neutrinos-Quarks-Inconsis.pdf —Preceding unsigned comment added by 97.77.60.254 (talk) 14:43, 6 October 2010 (UTC)
- What do you mean by "observed"?? Has an electron ever been observed? I've never seen one. Has the inside of a brick ever been observed? If you break one in half, you're still seeing the outside of both halves. You never will see the inside. So how do you know it exists at all? SBHarris 21:40, 6 October 2010 (UTC)
- The supporting evidence for the quark model is summarized at Quark#History. Perhaps this proton article could link to that section for readers who want to know why most physicists now accept that protons are made of quarks. Dirac66 (talk) 23:39, 6 October 2010 (UTC)
- Obviously Observered is a typo, which any native English speaker can recognize and understand as Observed. To dwell on spelling mistakes instead of answering the question is a logical fallacy called Ad Hominum. I understand the history of the quark, but that doesn't answer my question -- That is like saying The earth is flat, and if you don't believe it's flat simply read Aristotle and you will understand —Preceding unsigned comment added by 97.77.60.253 (talk) 18:11, 23 October 2010 (UTC)
- Sorry, I had missed the fact that "observed" was misspelled myself. Why don't you look at the rest of what I wrote, instead of making the mistake of being dragged into details, that you accuse me of? Do you think an electron has ever been observed? In the 25 years between when the neutrino was deduced as a way to save conservation of energy, momentum, and angular momentum in beta decay, and when it was "detected" by a certain expected reaction that occurred hear a nuclear reactor, do you think it existed? Have you ever seen one? Why talk about something you've never directly detected? What does "directly detected" MEAN, anyway? When high energy electrons are used to probe the structure of protons, it's clear that they're bouncing off three very small and "hard" objects inside. Not four, and not two. Only three explains the data. What do you want to call these "part-ons"? Look at the article on mesons. Mesons are understood to the point that most new ones have been predicted before the fact, like new elements from the periodic table. That means that mesons, like atoms, have a substructure that governs their properties. They are made of particles. What do you want to call them? SBHarris 18:22, 23 October 2010 (UTC)
- Your being rediculous, Why don't you do an experiment? -- Why don't you put your finger into an electrical socket so you will clearly find out that an electron exists???? Difference between what I say, and what you claim --Is that regardless of what the structure of the electron is, I can clearly prove it exists whereas the claim of a Quark is ludicrous under the assertion that individualy it cannot exist seperate of 3 other quarks Confinement of Quarks and what not. Maybe the Math for the Quantum physics of an electron is absolutely absurd but I can prove it exists.
All I see in everything I've ever read about the quark was that 3 is needed for the math to work right,not that a quark has ever been shown to exist, otherwise the data explodes. I do know of what your talking about regarding the nuclear reactor experiment, but I also realize that if you read the link that I posted (In my original question), which has now vanished for whatever reason, that scientists can explain the energy spike in Nuclear reactions without the need for the Quark. Which is why I asked the question in the first place http://www.i-b-r.org/Neutrinos-Quarks-Inconsis.pdf
Now if you could post links for the data, the research maybe a timeline or something of the various theories, regarding the quark, That would greatly help . Maybe even a University lecture or something clearly showing the MATH for the proof of the Quark, and how it explains things better than alternative theories being proposed then I wouldn't be here arguing with you. Thanks
- You could pick up a textbook on particle physics, like Quarks and Leptons, Francis Halzen and Alan D. Martin, Wiley and Sons, 1984. ISBN 0-471-88742-2 Parameter error in {{ISBN}}: checksum. It's more than 25 years old, but physics hasn't progressed all that much in that time, to tell the truth. The Standard Model (quarks) is the last good big theory that actually works. Chapter 9 focuses on the inelastic scattering patterns of electron-proton interactions, with various proton models. There are 3 hard objects (point-like Dirac particles) in there, that the electrons (actually, virtual photons from them) scatter off.
And by the way, sticking my finger in a socket tells me nothing about the existence or nonexistence of "electrons" -- it only shows there's "electricity" (access to an electric field via conductor). The field is the unpleasant part. People were making perfectly good electrical generators, and lighting electric bulbs with them, long before the electron was known. So were Maxwell's equations and primitive radio. If you care to Google, you'll find that the first man died in the electric chair 7 years before the electron was "discovered." What do you think they thought they were were doing to the poor guy? SBHarris 08:17, 24 October 2010 (UTC)
- You could pick up a textbook on particle physics, like Quarks and Leptons, Francis Halzen and Alan D. Martin, Wiley and Sons, 1984. ISBN 0-471-88742-2 Parameter error in {{ISBN}}: checksum. It's more than 25 years old, but physics hasn't progressed all that much in that time, to tell the truth. The Standard Model (quarks) is the last good big theory that actually works. Chapter 9 focuses on the inelastic scattering patterns of electron-proton interactions, with various proton models. There are 3 hard objects (point-like Dirac particles) in there, that the electrons (actually, virtual photons from them) scatter off.
- As a general note, Wikipedia is not a forum. If you don't understand quarks and neutrinos, pick up books on them, and stay away from Santilli's ludicrous writings. Anyone who writes
By the middle of the 20-th century there was no clear experimental evidence acceptable by the scientic community at large confirming the neutrino conjecture beyond doubt, except for experimental claims in 1959 that are known today to be basically flawed on various grounds, as we shall see in the next sections.
(in reference to the Cowan-Reines neutrino experiments which was in 1956 and not 1959 as he wrote), or
To begin, both neutrinos and quarks cannot be directly detected as physical particles in our spacetime. Consequently, all claims on their existence are indirect, that is, based on the detection of actual physical particles predicted by the indicated theories. This occurrence is, per se, controversial.
(in spite of the several dozens of direct neutrino detection experiments, or that the case for quarks is about as solid as it gets even if you'll never see the trace of a single lone quark in a bubble chamber) or
In the 1980s, a large laboratory was built deep into the Gran Sasso mountain in Italy to detect neutrinos coming from the opposite side of Earth since the mountain was used as a shield against cosmic rays. Following the investment of large public funds, the Gran Sasso Laboratory released no evidence of clear detection of neutrino originated events following five years of continuous tests.
(despite the very prolific record of the Grand Sasso Labs, and their several contributions to neutrino science (of the top of my head there was BOREXINO, CUORE, CUORICINO, GNO, GALLEX, MACRO, Heidelberg–Moscow, and many more) is disconnected from reality, either by ignorance or by the same mental disorder as the Flat Earth Society members have with regards to Earth's shape.
- As a general note, Wikipedia is not a forum. If you don't understand quarks and neutrinos, pick up books on them, and stay away from Santilli's ludicrous writings. Anyone who writes
- Ask yourself what's more likely. A) Each of the tens of thousands of scientist in the field is part of a conspiracy to prevent the "truth" about neutrinos and quarks to get out. They will build several billion-dollar facilities for the sole purpose of maintaining an illusion to fool the general public, and will dedicate their lives and countless hours of work to maintain that illusion. Not one of the several independent research groups will be willing to rise to international prominence by blowing the lid on this conspiracy, and only Santilli possess the courage to stand to the "scientific establishment". B) Santilli is wrong. Headbomb {talk / contribs / physics / books} 16:35, 24 October 2010 (UTC)
According to the article:
- "Protons are stable with an empirically observed half-life of at least 6.6×1035 years.[1] Although experiments so far have only resulted in a lower limit of 1033 years for the proton's lifetime, Grand unified theories generally predict that proton decay should take place. In other words, proton decay has never been witnessed and the experimental lower bound on the mean proton lifetime (2.1×1029 years) is given by the Sudbury Neutrino Observatory.[2]"
1. It says that protons are stable, then goes on to imply that it's not actually known if they are stable.
2. Three different figures are given for the lower bounds on the proton's "half-life", "lifetime", and "mean lifetime". It's not clear what "lifetime" means. As far as I understand it, half-life determines mean lifetime and vice versa, but the article makes it sound like "mean lifetime" is a separate and independent measure. Are the figures for "half-life" and "mean lifetime" mutually consistent?
3. The phrase "In other words" seems misplaced. 86.173.36.118 (talk) 23:24, 1 December 2010 (UTC).
- The word "stable" MEANS by definition that if it decays, the lifetime is so long that it's never been seen. Proving something is stable is as impossible as proving that it's immortal (same problem, actually). All you can do is prove that it isn't. Or that if it isn't, the lifetime must be longer than some minimum time.
This problem comes up a lot in the area of stable nuclides, where all nuclides of all elements after zirconium (#40) are theoretically unstable. But the lifetimes are so much longer than the lifetime of the universe, that nobody has measured a decay time. So we DEFINE something that is so long that we can't measure it, to be "stable." It means "stable for all intents and purposes, and to all present measurement." It's an empirical/experimental statement, not a philosophical one. It's subject to change with new evidence. See bismuth-209 for an example.
Mean lifetime = lifetime is longer than half life (see that article) by the factor 1/ln2 = 1.4427... This isn't a number large enough that it makes a difference if order-of-magnitude estimates are expressed as mean life or half life. So they can be used as interchangable terms. Pick which one you like. SBHarris 00:06, 2 December 2010 (UTC)
- Oh, OK, thanks, your explanation of "stable" makes sense. A couple of words in the article to that effect would not go amiss. I still don't understand what the figure for "lifetime" means, since only statistical measures such as mean lifetime or half-life appear to be meaningful. If I read you correctly, you seem to be implying that the figures in the article for the lowest known bounds on half-life and mean lifetime are greatly inconsistent. It's not really up to me to "pick which one I like". Ideally it should be the article's responsibility to account for the discrepancy. There also seems no need here to use two different ways of quoting what is fundamentally the same quantity. 86.173.36.118 (talk) 01:04, 2 December 2010 (UTC).
- "Lifetime" is (strictly speaking) wrong for a collection of particles, since it implies a non-statistical concept (valid only for ONE particle, and measurable when it decays). "Mean-lifetime" is the term that is always meant for collections, when somebody says "life" but not "half life". Mean life is the "1/e life," rather than the "1/2 life" (half life).
The various different figures in the article are due to various different experimental ways of setting a lower bound for proton half-life (or mean-life which is 44% longer), or calculating it from theory, not due to the difference between half-life and mean-life, which (as discussed) is too small to show in these numbers. One or the other of these should be picked and used throughout, with perhaps the other used just once. This is done in the article on the neutron, for example, where mean-life is used throughout, but half-life is given once. SBHarris 01:21, 2 December 2010 (UTC)
- "Lifetime" is (strictly speaking) wrong for a collection of particles, since it implies a non-statistical concept (valid only for ONE particle, and measurable when it decays). "Mean-lifetime" is the term that is always meant for collections, when somebody says "life" but not "half life". Mean life is the "1/e life," rather than the "1/2 life" (half life).
- Oh, OK, thanks, your explanation of "stable" makes sense. A couple of words in the article to that effect would not go amiss. I still don't understand what the figure for "lifetime" means, since only statistical measures such as mean lifetime or half-life appear to be meaningful. If I read you correctly, you seem to be implying that the figures in the article for the lowest known bounds on half-life and mean lifetime are greatly inconsistent. It's not really up to me to "pick which one I like". Ideally it should be the article's responsibility to account for the discrepancy. There also seems no need here to use two different ways of quoting what is fundamentally the same quantity. 86.173.36.118 (talk) 01:04, 2 December 2010 (UTC).
- The problem is that there are 3 different values given for the experimental lower bound, of the order of 1036, 1033 and 1029 yr. As you say, the factor of ln 2 is close to 1 and is insignificant here, so essentially we have 3 widely differing values for the same quantity in the same section. If this is due to "various different experimental ways of setting a lower bound", the difference between the three experiments needs to be explained.
- In fact I asked this question exactly 2 years ago (see above section 22.Lifetime) when there were only 2 values in the article. Blennow answered then that one value was for "visible" decays and the other for decays to "anything". I did not and do not understand this very well, so I asked a followup question (see above) which was never answered. If you can explain the difference between the 3 values, and identify which value is most meaningful, that would be very good. (I would normally guess that the highest lower bound is best, IF the experiments are equally reliable.) Dirac66 (talk) 01:38, 2 December 2010 (UTC)
Just noticed that the 6.6x1035 yr value is 6.6x1033 in the proton decay article, from the same source. So I checked Phys Rev Letters, and it is 6.6x1033 (for the partial lifetime via antimuon decay). I have now corrected this article - checking sources sometimes helps. Dirac66 (talk) 02:08, 21 December 2010 (UTC)
Here I mean "ordinary" as on Earth at STP, not in the cores of neutron stars. No, free protons do not exist in liquids, since they always attach themselves to the electron cloud in the nearest molecule/anion, which is always available. There may exist free "solvated electrons" in liquids (like sodium dissolving in liquid ammonia to give a nice color), but electrons have sqrt(1836) = 43 times longer wavelengths than do protons, at any energy. A proton at room temp has a wavelength of 2 angstroms or so, which is far too small for them to sit in a cage between negatively charged anions and not be able to make a choice of which way to go and which to sit on, like the proverbial donkey starving between piles of hay. Protons have no reason to delocalize at room temperature.
Yes, I know the article on superacids talks about free protons in liquids, but it gives no references, and frankly I don't believe it. Ab initio QM calculations show protons hopping from anion-to-anion in the strongest superacid known, just as they do in water, via the Grotthuss mechanism. See .
So, some editor who claims to know what he's talking about, has reverted me on this point. There's my cite and there's my reasoning. Now, what have YOU got? And by the way, I'm going to go add a [citation needed] to the statement in superacids. SBHarris 00:36, 27 February 2011 (UTC)
- De Broglie wavelength of the proton apparently is something on topic, but such waves are relevant to free motion only, or if the force potential changes on far larger scale, isn't it? I do not see (yet) something that contradicts to the existence of a proton quite weakly bounded to nearby molecules (of course, not absolutely free, even molecules in a real gas are not free). Also, no clarifications about proton's alleged strong electrostatic field were provided. May be, we should first develop a consensus to what extent should this controversy described in this article? Incnis Mrsi (talk) 18:21, 27 February 2011 (UTC)
- De Broglie wavelengths are always relevant, any time a particle has a momentum: λ = h/p. The de Broglie wavelength for bound electrons is what determines the size of ordinary atoms.
Whether a particle is "bound" or "free" is little bit of a semantic quibble, but if a proton is always more closely connected to any one cation or two cations, than it is to any others, it makes sense to say that it's bound to them. It may be free to wander through a liquid, but only by going from one molecule to another, and being passed off from one to another like a prisoner being transferred betwee guards, always handcuffed to at least one at a time (sometimes to two, during a transfer). The article shows that happening in the liquid in which protons are most "free" of all (the strongest acid) so it certainly must be true in all other ordinary liquids at STP. And we can see with anions compounds like FHF- and FHFHF- how badly a hydrogen wants to be connected to, or sandwiched between, fluorines. It doesn't just disconnect from one fluorine and go wandering through the liquid as a naked proton, before it finds another. That's a more "wrong" physical picture than the reality. That kind of thing apparently does happen in hydrogen plasmas, but the temperature there is far higher than the binding energies of electrons to bare-protons (13.6 eV vs. about 0.025 eV at room temp). As for the electric field, and more importantly, the potential, at various distances from a proton, you can calculate it. You tell me the binding energy of an fluorine electron to a fluorine, and I will tell you how close one must get to a proton to exceed that (with a naked proton there is no limit), and THEREFORE how close a proton needs to come to a fluorine before the fluorine must surrender (at least in part) one if its electrons. This is very simple and there's no getting around it. You can think of a proton as having an electron affinity of -13.6 eV, which is far beyond any other chemical bond energy. A proton WILL approach a fluorine until it binds. A proton will approach ANY ion that that has electrons, until it binds. See "The Proton Lewis Acid" in . I'll just stop here and let you answer. SBHarris 17:42, 18 March 2011 (UTC)
- Taking the charge radius, r, of the proton as ca. 0.88 fm (from proton) and comparing it with Li+ at 60 pm, the charge to size ratio of the bare proton is some 70000 times larger than the next smallest cation. It is therefore such a very, very strong Lewis acid that its lifetime in any chemical system will be extremely short. I can imagine that there might be some data in the field of pulse radiolysis on the reactivity of the bare proton, but otherwise it is effectively non-existent in chemical systems. Petergans (talk) 08:05, 19 March 2011 (UTC)
- De Broglie wavelengths are always relevant, any time a particle has a momentum: λ = h/p. The de Broglie wavelength for bound electrons is what determines the size of ordinary atoms.
Just a little clarification. Where it is necessary in chemistry to distinguish between 1H and 2H it is usual to use the terms proton and deuteron, denoted by H and D. I am not aware of any similar name for 3H, T. Petergans (talk) 11:45, 18 March 2011 (UTC)
- The word triton exists in nuclear physics, but is admittedly very rare in chemistry. A Google search for (triton + deuteron) yields 158 K hits, the first few mostly in nuclear physics. A Google search for just triton yields 25 M hits, the first few mostly wholly irrelevant since the word has many meanings. Dirac66 (talk) 13:55, 18 March 2011 (UTC)
| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 1 |
To Krebs49, who attempted today to add the magnetic shielding correction value to the infobox and noted in his edit summary that the added value does not appear: I believe the problem is that the infobox is generated using Template:Infobox particle and can only show the parameters listed there. So you could either (1) edit the template to include Magnetic shielding correction (and carefully check that this action does not otherwise affect either this Proton article or the articles for other particles), or (2) leave the infobox alone and put the magnetic shielding correction into the text somewhere. Dirac66 (talk) 20:02, 3 April 2014 (UTC)
I've been poking at the article for neutron of late, with occasional visits to this page. The two articles (and likely others) should have a degree of similarity about them, seems to me. The History section on this article is rather far down in the article, whereas it is at top in the neutron article. I tend to think the history section should be toward the top since this section also serves as an introduction to the topic. In any case, a certain uniformity in article design would likely be helpful. Bdushaw (talk) 03:19, 15 September 2014 (UTC)
- I consider that a synchronization of this article with electron, beside that with neutron, is useful and recommended.--85.121.32.27 (talk) 11:38, 19 September 2014 (UTC)
The three-balls-held-together-with-springs illustration is, apparently, an oversimplified model of what is inside a proton, which has been outdated since the 1970's. However, a more accurate model would be, apparently, quite complex-- see this illustration: [[File:MyProton|thumbnail|Prof Matt Strassler's illustration of a proton - complex]] and the article from which it comes: http://profmattstrassler.com/articles-and-posts/largehadroncolliderfaq/whats-a-proton-anyway/ I suggest that we put both the simplified, iconic image, and the less-simplified image, suitably captioned.
I don't know if Matt Strassler approves his image for use on Wikipedia or not. Dc3 (talk) 19:28, 4 June 2014 (UTC)
- I do not see any point in making the figure more complicated by trying to depict the sea quarks. I think it is still completely valid to say that the proton is made of three (valence) quarks. The "sea quarks" arise from the strong interaction itself, which is depicted with the wavy lines. In a sense, these waves include the sea quarks. If one goes down the path of depicting the sea quarks, why not also the virtual photons, electrons, W bosons, etc. The figure becomes very confusing. 128.141.41.38 (talk) 10:21, 16 March 2015 (UTC)
The occurence/existence of any compounds which contain bare proton as cation would be a useful addition to article. Is there any awareness of sources with such compounds described?--79.119.209.7 (talk) 11:20, 8 November 2015 (UTC)
The article says that a quantity called classical radius of a charged particle is convenient to be defined. Can also proton have a classical radius defined for for it?--5.2.200.163 (talk) 13:51, 12 February 2016 (UTC)
The radius given in the body of the text is given as 0.84–0.87 fm, whereas in the top-right table it is given as 0.8775(51) fm. I understand that the former one is due to a recent discovery of a different radius and the top-right table is based on a table published prior to that study, but it will be confusing to readers. A more recent data table for the proton (2014), from the particle data group rather than CODATA, gives both values: http://pdg.lbl.gov/2014/listings/rpp2014-list-p.pdf I suggest doing the same in the top-right box, using this as a reference, and maybe add some details on the controversy to the body of the text. MostlyForgettable (talk) 21:23, 26 March 2015 (UTC)
- What controversy? You mean discrepancy?--188.26.17.81 (talk) 19:03, 7 November 2015 (UTC)
- What's the difference? 128.135.100.105 (talk) 04:08, 6 October 2016 (UTC)
Measurement details
I see above that values of the proton's radius are mentioned. I think it would be useful also that details of measuring methods and their principles be inserted in article.--213.233.84.189 (talk) 23:52, 30 December 2015 (UTC)
- I understand that scattering data are used to determine the radius of proton. Is it a direct measurement or scattering data are introduced in a formula?--213.233.84.189 (talk) 00:58, 31 December 2015 (UTC)
- I believe that the formula you asked about was inserted into the article, but it seems insufficiently melded with the surrounding text. I don't know enough QFT to understand how these form factors are useful. Should they be here, or in the main page on the proton radius puzzle? If they should be here, can you improve it? Thanks, 128.135.100.105 (talk) 04:08, 6 October 2016 (UTC)
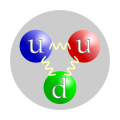
The first image on the page INCORRECTLY denotes the quark structure of a proton. The gluon fields binding the the three quarks together should not shown as connecting one quark to another and forming a triangular shape; rather, the gluon fields should be originating from the direct center of the image and extending outward to the quarks to form a Y-like shape. 67.61.85.45 (talk) 22:13, 5 July 2013 (UTC)
- I do not think so. See asymptotic freedom. Incnis Mrsi (talk) 05:21, 6 July 2013 (UTC)
Incnis: citing another Wiki article is no justification whatsoever. I have just watched a YouTube video which also explicitly makes the claim that the diagram is wrong, and the person making this claim is, according to the video, Professor Derek Leinweber of Univ. of Adelaide. https://www.youtube.com/watch?v=Ztc6QPNUqls&feature=player_embedded This directly and explicitly discusses that diagram and says (3 min 30 second into video) that we now know that it is wrong. The diagram needs to be fixed or removed.72.172.11.140 (talk) 20:48, 29 January 2014 (UTC)
- Are sand–PVC models, some speaking faces, and their strangely distorted speech (something like “We know that all picture is totally wrong now. Even if… blah-blah… would see quark tubes around… blah-blah-blah-blah… more than three quarks…”) all that you can present? Give a citation, please, exactly on the question of Y-shape vs triangle. Possibly from the same Derek Leinweber, but as a legible text. Incnis Mrsi (talk) 12:53, 30 January 2014 (UTC)
- The video has a link to the man's website in the description, which itself links back to the video, here: http://www.physics.adelaide.edu.au/theory/staff/leinweber/ On that page is a link to another page, here: http://www.physics.adelaide.edu.au/theory/staff/leinweber/VisualQCD/Nobel/index.html which includes a gif showing the Y-shaped flux tubes of three protons of different colours, here: http://www.physics.adelaide.edu.au/theory/staff/leinweber/VisualQCD/Nobel/VacuumRespAction16t32_Yshape8small.gif Would it be possible to just use that gif in the wiki article, actually? MostlyForgettable (talk) 20:49, 26 March 2015 (UTC)
I mean, if you really wanted to be accurrate, instead of wavy lines infinitely many quarks and gluons should be drawn. But when you get down to that level of scale any diagram will be inaccurate in some way or another (how would you represent uncertainty, for instance). The one that is currently there works just fine in getting the general point across. ArchPope Sextus VI (talk) 01:48, 15 January 2015 (UTC)
- I agree that the diagram was never intended to present things in infinite detail, since doing so is impossible in principle because of Heisenberg's principle and quantum electrodynamics. Such diagrams are only intended to be rough approximations, and arguments about "triangle shaped" versus "Y - shaped" are neither here nor there. In other words, they are specious.47.215.211.115 (talk) 10:35, 12 January 2017 (UTC)
The rest mass of a proton is about 1836 times the rest mass of an electron. This is an experimental fact, and it does not have a theoretical basis.
This very important physical constant had not even been measured before I added it tonight. Also, on the subatomic level, the mass of the electronic is a very important unit of mass, and a fundamental one to the physical Universe.47.215.211.115 (talk) 10:42, 12 January 2017 (UTC)
A new experiment measured the mass of a proton at 1.007276466583 atomic mass units, see here. --167.58.203.60 (talk) 13:18, 21 July 2017 (UTC)
- Not very helpful if they don't provide the uncertainty as well. We need to wait for others to validate the results anyway. Sizeofint (talk) 17:58, 24 July 2017 (UTC)
- current version
- proposed new version
This particular picture from Wikipedia is proven wrong mathematically. Details: http://backreaction.blogspot.co.uk/2017/12/get-your-protons-right.html 185.20.63.25 (talk) 15:56, 18 December 2017 (UTC)
- Fixed. Olaf (talk) 22:22, 14 January 2018 (UTC)
- The change was reverted by user:Headbomb with comment "get consensus for this first". Well, the current image is wrong because the strong force between quarks is not a two-particle relationship, and this particular Y-shape configuration was derived in this paper: . For further discussion see . If there will be no physical reasons against it in the next week, I'm going to change it again. Olaf (talk) 00:19, 15 January 2018 (UTC)
- Would it be possible to explain simply in this article what is implied by the proposed change of diagram? My uninformed guess is that it means there is no force between 2 quarks unless a third is present. If my guess is wrong, then please tell the reader what it does mean. If my guess is right, then can you briefly summarize the evidence that 3 quarks are required for a strong force? Dirac66 (talk) 00:45, 15 January 2018 (UTC)
- There is no such thing like two isolated quarks (see color confinement), and in a three-quark system you cannot calculate the strong force as a sum of forces between each pair of quarks (as was possible in case of the classic electromagnetism or gravity). Yes, there is a strong force between a quark and an antiquark in a meson, and it doesn't need the third particle, but it is irrelevant. In fact this diagram tries to show something different – the gluon-fluxes between quarks. It was derived mathematically, that gluons in a proton don't form a triangle, but rather a dynamically changing Y-shape. See: . Olaf (talk) 01:14, 15 January 2018 (UTC)
- Would it be possible to explain simply in this article what is implied by the proposed change of diagram? My uninformed guess is that it means there is no force between 2 quarks unless a third is present. If my guess is wrong, then please tell the reader what it does mean. If my guess is right, then can you briefly summarize the evidence that 3 quarks are required for a strong force? Dirac66 (talk) 00:45, 15 January 2018 (UTC)
- The change was reverted by user:Headbomb with comment "get consensus for this first". Well, the current image is wrong because the strong force between quarks is not a two-particle relationship, and this particular Y-shape configuration was derived in this paper: . For further discussion see . If there will be no physical reasons against it in the next week, I'm going to change it again. Olaf (talk) 00:19, 15 January 2018 (UTC)
- The diagrams depict different things. This is an extremely simplified conceptual diagram showing that quarks interact with each other via gluon exchange, not a representation of the physical layout of gluon flux tubes, which is shaped by quark-gluon interactions, as well as gluon-gluon interactions, and involves both sea quarks and valence quarks. We could decide to switch to diagrams with gluon flux tubes instead, but I'm not sure the reader benefits from this.
- If the diagram is so simplified, that it doesn't matter where the lines are drawn, the new version can symbolize the quark-quark interaction too, being at the same time more accurate. And it's not the quark-quark interaction. It's a quark-quark-quark interaction. Olaf (talk) 01:20, 15 January 2018 (UTC)
- This also would affects more than just protons, every concept diagram of baryons would be affected by this as well. Headbomb {t · c · p · b} 00:53, 15 January 2018 (UTC)
- In fact only two diagrams - of a proton and a neutron should be changed. Olaf (talk) 01:20, 15 January 2018 (UTC)
- There's also antineutron, antiproton, pentaquark, and File:Quark structure omega.svg, and would also affect any images being created for say Lambda baryon and the like. Headbomb {t · c · p · b} 01:28, 15 January 2018 (UTC)
- The first three can be easily changed, but I admit – the corrected diagrams are not made yet. The pentaquark diagram is totally out of line – it's captioned "The wavy lines are gluons, which mediate the strong interaction between the quark" and again – the gluon flux is unlikely to look like that, because the gauge invariance would be violated just like in the proton. Or is it just an iteraction diagram, wrongly captioned? Then could you show me the scientific source of the decision which quarks are connected on the pentaquark diagram? I'm sorry, but it looks like an OR – from this picture you can see, that each quark interacts with each other. Olaf (talk) 01:42, 15 January 2018 (UTC)
- That's because you insist on seeing those as gluon flux tubes when that's not what those lines represent. Headbomb {t · c · p · b} 01:56, 15 January 2018 (UTC)
- I've just read the caption in the pentaquark: "The wavy lines are gluons"... Still, if the lines symbolize just interaction between quarks, not gluons, the diagram is wrong too, because you can't treat interaction between two quarks in the isolation from the rest of the system, and all five quarks in the pentaquark interact with each other which is inaccurately depicted in the current diagram. So the star-shaped diagram is more accurate even if you insist we don't draw the gluon flux tubes. Olaf (talk) 02:09, 15 January 2018 (UTC)
- It's a simplified diagram to illustrate the concept, it is not a 1:1 representation of every possible interaction going on. For that you'd need to add sea quarks, quark-quark interactions, gluon-gluon interactions, quark-gluon interactions, and a bunch of other things. Headbomb {t · c · p · b} 03:19, 15 January 2018 (UTC)
- The same concept can be as well ilustrated by the new image, which is less controversial, and not criticized by scientists. The only advantage of the old diagram is that it matches the same style in other diagrams. But the other diagrams are being converted too. Olaf (talk) 23:42, 18 January 2018 (UTC)
- It's a simplified diagram to illustrate the concept, it is not a 1:1 representation of every possible interaction going on. For that you'd need to add sea quarks, quark-quark interactions, gluon-gluon interactions, quark-gluon interactions, and a bunch of other things. Headbomb {t · c · p · b} 03:19, 15 January 2018 (UTC)
- I've just read the caption in the pentaquark: "The wavy lines are gluons"... Still, if the lines symbolize just interaction between quarks, not gluons, the diagram is wrong too, because you can't treat interaction between two quarks in the isolation from the rest of the system, and all five quarks in the pentaquark interact with each other which is inaccurately depicted in the current diagram. So the star-shaped diagram is more accurate even if you insist we don't draw the gluon flux tubes. Olaf (talk) 02:09, 15 January 2018 (UTC)
- That's because you insist on seeing those as gluon flux tubes when that's not what those lines represent. Headbomb {t · c · p · b} 01:56, 15 January 2018 (UTC)
- The first three can be easily changed, but I admit – the corrected diagrams are not made yet. The pentaquark diagram is totally out of line – it's captioned "The wavy lines are gluons, which mediate the strong interaction between the quark" and again – the gluon flux is unlikely to look like that, because the gauge invariance would be violated just like in the proton. Or is it just an iteraction diagram, wrongly captioned? Then could you show me the scientific source of the decision which quarks are connected on the pentaquark diagram? I'm sorry, but it looks like an OR – from this picture you can see, that each quark interacts with each other. Olaf (talk) 01:42, 15 January 2018 (UTC)
- There's also antineutron, antiproton, pentaquark, and File:Quark structure omega.svg, and would also affect any images being created for say Lambda baryon and the like. Headbomb {t · c · p · b} 01:28, 15 January 2018 (UTC)
- In fact only two diagrams - of a proton and a neutron should be changed. Olaf (talk) 01:20, 15 January 2018 (UTC)
- The diagrams depict different things. This is an extremely simplified conceptual diagram showing that quarks interact with each other via gluon exchange, not a representation of the physical layout of gluon flux tubes, which is shaped by quark-gluon interactions, as well as gluon-gluon interactions, and involves both sea quarks and valence quarks. We could decide to switch to diagrams with gluon flux tubes instead, but I'm not sure the reader benefits from this.
See also Talk:Proton#Inaccurate information and Talk:Proton#Inaccurate illustration above, as well, for similar discussions. Headbomb {t · c · p · b} 00:57, 15 January 2018 (UTC)
Theorized by Prout in XIX century?--Agremon (talk) 15:23, 9 December 2018 (UTC)
As of 20 May 2019, the charge of a proton is exactly 1.602176634e-19 C. This article needs to be updated accordingly. See https://en.wikipedia.org/wiki/2019_redefinition_of_the_SI_base_units David Boyd Hansen (talk) 18:43, 22 August 2019 (UTC)

An editor has asked for a discussion to address the redirect Antinegatron. Please participate in the redirect discussion if you wish to do so. –LaundryPizza03 (dc̄) 07:06, 25 April 2020 (UTC)
The article says: (the values in parentheses being the statistical and systematic uncertainties, respectively), except that there aren't parentheses, except around that statement. Gah4 (talk) 21:20, 26 June 2020 (UTC)
I recently noted that the mass listed in the infobox on the deuterium page includes the mass of the electron. (This caused me an hour of puzzlement, but nevermind.). The infobox is frankly confusing since it seems to be an isotope infobox, but then it lists nuclide data, giving the mass of the isotope (which I would take to include the electron, maybe). I'm not sure how to resolve the issue, and my Talk entry on the deuterium page has gone unanswered - further down in the deuterium article the bare mass is given. I write here because it raises a general question on how the masses of nuclides are listed - it seems to me that both the masses of the particle with and without the electron(s) should be listed. This article, for the proton, lists the mass without the electron, but does not give the mass of the proton with the electron. I thought about editing the infobox for the isotope to have it give entries for both nuclide and isotope, but I'm not sure how to do that, or if that is even the correct way of doing it. Or is this issue just particular to hydrogen/deuteron - that is, does the mass of the carbon nuclide include the mass of the 12 electrons? Mass confusion, as I say... Bdushaw (talk) 10:44, 23 December 2017 (UTC)
- From my point, the mass of the hydrogen and deuterium should be with the mass of the electron. If you ask for the mass of the proton and the alpha-particle it should be without electron. But for an chemist the mass of the electron does not matter most of the time. --Stone (talk) 16:52, 23 December 2017 (UTC)
- The proton, deuteron and C-12 nuclEUS are bare nuclei and do not include electrons. The hydrogen atom, deuterium atom and C-12 nuclIDE (or isotope) are atoms and do include electrons. Dirac66 (talk) 18:27, 23 December 2017 (UTC)
- I understand the difference (though personally, I would have thought nuclides would exclude the electron, while isotopes would include them)...I suppose I am objecting to the lack of a consistent listing of the masses on the various pages; I am suggesting a consistent approach would be better. Over on the Hydrogen page, both masses are listed, but without an indication as to why two masses are listed! I anticipate that masses with the electron would also have to include the negative contribution of the small electron binding energy (13.6 eV in the case of hydrogen). When one says the mass of the carbon 12 nuclide is exactly 12, by definition, that most likely does not include the 12 electron masses/binding energies of the carbon atom. Bdushaw (talk) 22:47, 26 December 2017 (UTC)
- The proton, deuteron and C-12 nuclEUS are bare nuclei and do not include electrons. The hydrogen atom, deuterium atom and C-12 nuclIDE (or isotope) are atoms and do include electrons. Dirac66 (talk) 18:27, 23 December 2017 (UTC)
- Re inconsistency: This is part of the nature of Wikipedia, where different articles are written independently by different editors. You could of course go through all the pages of a given type and impose consistency, but that would be a lot of work and future editors might not respect your finished product.
- Re mass of carbon 12: Remember that u is the unified atomic mass unit which was defined in 1961 as one twelfth of the mass of an unbound neutral atom of carbon-12. So yes, the mass of such an atom is exactly 12 by definition. Dirac66 (talk) 03:21, 27 December 2017 (UTC)
- I think I asked this on a page about nuclides some time ago, and it seems that the answer is that it does include the electron. Since nuclides are commonly used for nuclear physics, I might have thought that they didn't. If there was an article on the deuteron (I didn't check) that would be without an electron. Gah4 (talk) 21:23, 26 June 2020 (UTC)
- OK, it links here, as I would have guessed. (So maybe the page should also give the mass for the deuteron.) I also notice that it mentions deuterium in stars. Maybe that should be deuterons, as there are no free (unionized) atoms in the core of stars. Gah4 (talk) 21:26, 26 June 2020 (UTC)
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.