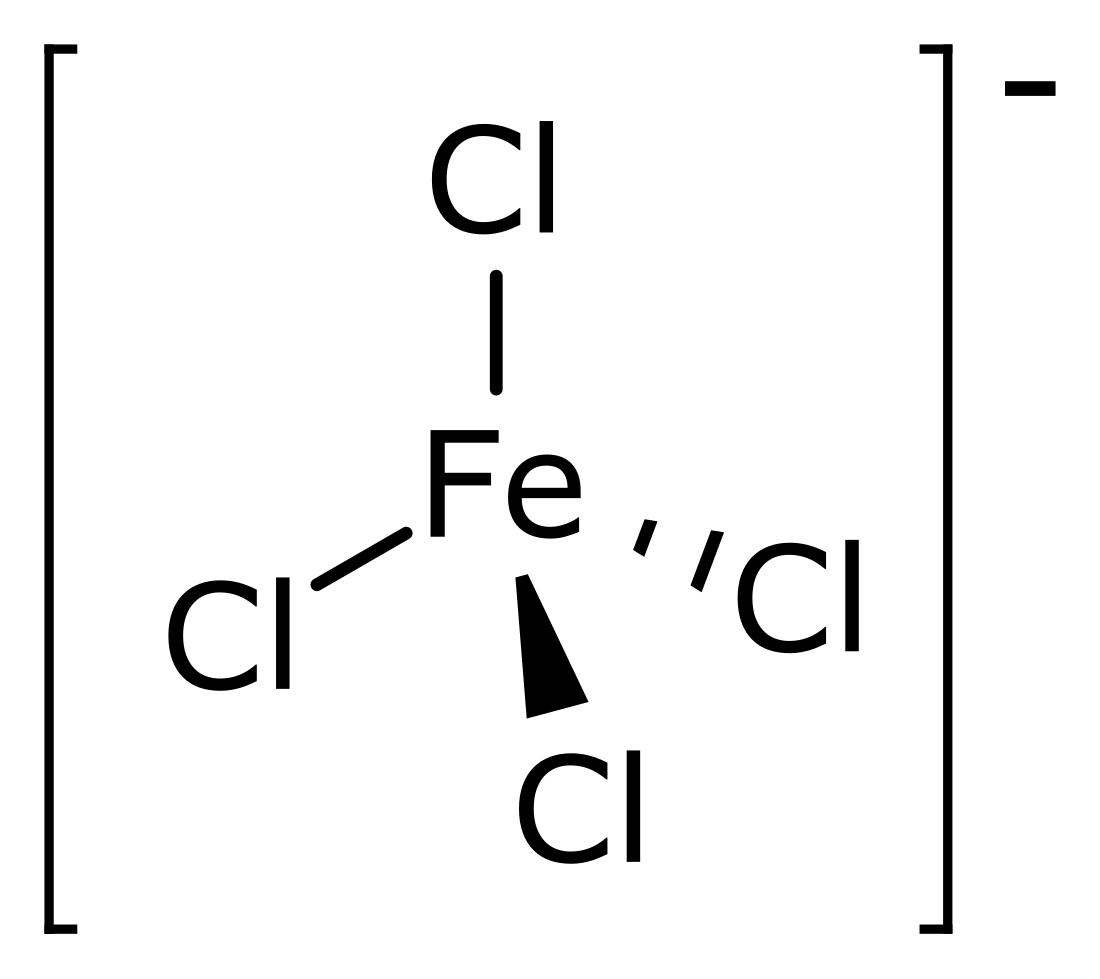
Tetrachloroferrate is the polyatomic ion having chemical formula FeCl−4. The metallate can be formed when ferric chloride (FeCl3) abstracts a chloride ion from various other chloride salts.[1] The resulting tetrachloroferrate salts are typically soluble in non-polar solvents. The tetrachloroferrate anion, with iron(III) in the center, has tetrahedral geometry.[2] It is useful as a non-coordinating anion comparable to perchlorate.[3] Several organoammonium salts have been studied for their novel material properties. 1-Butyl-3-methylimidazolium tetrachloroferrate is one of several ionic liquids that are magnetic.[4] Trimethylchloromethylammonium tetrachloroferrate is a plastic crystal that can behave as a molecular switch in response to several different types of inputs.[5]
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| 26231 | |
PubChem CID |
|
| |
| |
| Properties | |
| Cl4Fe−1 | |
| Molar mass | 197.65 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.