
Tetraethylammonium chloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tetraethylammonium chloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Tetraethylammonium chloride (TEAC) is a quaternary ammonium compound with the chemical formula [N(CH2CH3)4]+Cl−, sometimes written as [NEt4]Cl. In appearance, it is a hygroscopic, colorless, crystalline solid. It has been used as the source of tetraethylammonium ions in pharmacological and physiological studies, but is also used in organic chemical synthesis.
Quick Facts Names, Identifiers ...
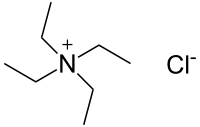 | |
| Names | |
|---|---|
| Preferred IUPAC name
N,N,N-Triethylethanaminium chloride | |
| Other names
Tetraethylammonium chloride N,N,N,N-Tetraethylammonium chloride | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.000.243 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| [N(CH2CH3)4]Cl | |
| Molar mass | 165.71 g·mol−1 |
| Appearance | Colorless deliquescent crystalline solid |
| Density | 1.08 g/cm3[1] |
| Melting point | 360 °C (680 °F; 633 K) tetrahydrate[1] |
| highly soluble | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
65 mg/kg (mouse, i.p.) 900 mg/kg (mouse, p.o.) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close