
Trofinetide
Pharmaceutical drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trofinetide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Trofinetide, sold under the brand name Daybue, is a medication used for the treatment of Rett syndrome.[1] It is taken by mouth.[1]
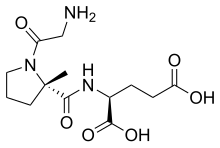
Quick Facts Clinical data, Trade names ...
 | |
| Clinical data | |
|---|---|
| Trade names | Daybue |
| Other names | NNZ-2566 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a623019 |
| License data |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 84% |
| Metabolism | Insignificant |
| Elimination half-life | ~ 1.5 h |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| Chemical and physical data | |
| Formula | C13H21N3O6 |
| Molar mass | 315.326 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Close
The most common adverse reactions include diarrhea and vomiting.[2]
Trofinetide was approved for medical use in the United States in March 2023.[1][2][3] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication.[4]