
Umifenovir
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Umifenovir?
Summarize this article for a 10 year old
Umifenovir, sold under the brand name Arbidol, is an antiviral medication for the treatment of influenza and COVID infections used in Russia[5] and China. The drug is manufactured by Pharmstandard (Russian: Фармстандарт). It is not approved by the U.S. Food and Drug Administration (FDA) for the treatment or prevention of influenza.[6]
You can help expand this article with text translated from the corresponding article in Russian. (June 2020) Click [show] for important translation instructions.
|
This article needs more reliable medical references for verification or relies too heavily on primary sources. (July 2020) |  |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Arbidol |
| Other names | AR-1I9514 |
| Pregnancy category |
|
| Routes of administration | By mouth (hard capsules, tablets) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40% |
| Metabolism | Hepatic, CYP3A4[3] |
| Elimination half-life | 17–21 hours |
| Excretion | 40% excrete as unchanged umifenovir in feces (38.9%) and urine (0.12%)[4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.247.800 |
| Chemical and physical data | |
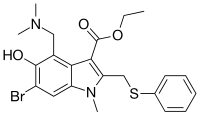
| Formula | C22H25BrN2O3S |
| Molar mass | 477.42 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Chemically, umifenovir features an indole core, functionalized at all but one positions with different substituents. The molecular groups of umifenovir - hydroxy, amino and carboxy - can interact to form various hydrogen-bonded synthons. Antiviral activity and acceptable cytotoxicity profiles [7][8] make it a promising candidate for further research as a potential therapeutic agent for the selective treatment of flavivirus infections [9]. Umifenovir is characterized by only one polymorphic form but can exist in the form of a large number of crystal solvates, the production of which depends on the medium and conditions of synthesis. In this case, the implementation of the crystal solvate or other solid form may be determined by the spatial structure and conformational equilibria in the saturated solution [10][11]. The drug has been shown in studies to inhibit viral entry into target cells [12] and stimulate the immune response.