
Vanadium(III) fluoride
Chemical compound / From Wikipedia, the free encyclopedia
Vanadium(III) fluoride is the chemical compound with the formula VF3. This yellow-green, refractory solid is obtained in a two-step procedure from V2O3.[4] Similar to other transition-metal fluorides (such as MnF2), it exhibits magnetic ordering at low temperatures (e.g. V2F6.4H2O orders below 12 K[5]).
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Other names
Vanadium fluoride, Vanadium trifluoride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.030.141 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| F3V | |
| Molar mass | 107.9367 g·mol−1 |
| Appearance | Yellow-green powder (anhydrous) Green powder (trihydrate)[1] |
| Density | 3.363 g/cm3[1] |
| Melting point | 1,395 °C (2,543 °F; 1,668 K) at 760 mmHg (anhydrous) ~ 100 °C (212 °F; 373 K) at 760 mmHg (trihydrate) decomposes[1] |
| Boiling point | Sublimes |
| Insoluble[1] | |
| Solubility | Insoluble in EtOH[1] |
| 2.757·10−3 cm3/mol[1] | |
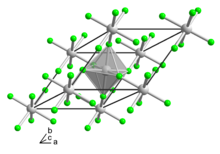
| Structure | |
| Rhombohedral, hR24[2] | |
| R3c, No. 167[2] | |
| 3 2/m[2] | |
α = 90°, β = 90°, γ = 120° | |
| Hazards | |
| GHS labelling: | |
  [3] [3] | |
| Danger | |
| H301, H311, H314, H331[3] | |
| P261, P280, P301+P310, P305+P351+P338, P310[3] | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions |
Vanadium(III) chloride Vanadium(III) oxide Vanadium(III) nitride |
Other cations |
Vanadium(IV) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
