Top Qs
Timeline
Chat
Perspective
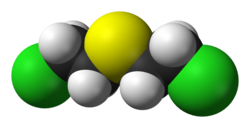
Bis(2-chloroethyl)sulfide
Chemical compound formerly used in warfare From Wikipedia, the free encyclopedia
Remove ads
Bis(2-chloroethyl)sulfide is the organosulfur compound with the formula (ClCH2CH2)2S. It is a prominent member of a family of cytotoxic and blister agents known as mustard agents. Sometimes referred to as mustard gas, the term is technically incorrect: bis(2-chloroethyl)sulfide is a liquid at room temperature. In warfare it was dispersed in the form of a fine mist of liquid droplets.[4][5]
Remove ads
Synthesis
Bis(2-chloroethyl)sulfide has been prepared in a variety of ways. In the Depretz method, sulfur dichloride is treated with ethylene:
- SCl2 + 2 C2H4 → (ClC2H4)2S
In the Levinstein process, disulfur dichloride is used instead:[6][failed verification][7]
- S2Cl2 + 2 C2H4 → (ClC2H4)2S + 1⁄8 S8
In the Meyer method, thiodiglycol is produced from chloroethanol and sodium sulfide, and the resulting diol is then treated with phosphorus trichloride:[8][9]
- 3 (HOC2H4)2S + 2 PCl3 → 3 (ClC2H4)2S + 2 P(OH)3
In the Meyer–Clarke method, concentrated hydrochloric acid (HCl) is used instead of PCl3:
- (HOC2H4)2S + 2 HCl → (ClC2H4)2S + 2 H2O
Thionyl chloride and phosgene, the latter of which is also a choking agent, have also been used as chlorinating agents. These compounds have the added advantage in that if they are used in excess, they remain as impurities in the finished product and can therefore produce additional mechanisms of toxicity.
Remove ads
Reactions
The idealized combustion of mustard gas in oxygen produces hydrochloric acid and sulfuric acid, in addition to carbon dioxide and water:
- (ClC2H4)2S + 7 O2 → 4 CO2 + 2 H2O + 2 HCl + H2SO4
Bis(2-chloroethyl)sulfide reacts with sodium hydroxide, giving divinyl sulfide:
- (ClC2H4)2S + 2 NaOH → (CH2=CH)2S + 2 H2O + 2 NaCl
Sodium ethoxide acts similarly.
Safety
Upon skin contact or inhalation, bis(2-chloroethyl)sulfide is a nonspecific toxin. It is a strong alkylating agent, which affects DNA, RNA, and proteins.[10]
See also
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads