Top Qs
Timeline
Chat
Perspective
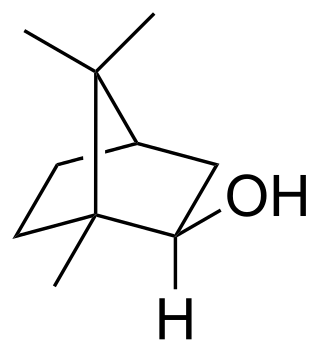
Isoborneol
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
Isoborneol is a bicyclic organic compound and a terpene derivative. The hydroxyl group in this compound is placed in an exo position. The endo diastereomer is called borneol. Being chiral, isoborneol exists as enantiomers.
Remove ads
Preparation
Isoborneol is synthesized commercially by hydrolysis of isobornyl acetate. The latter is obtained from treatment of camphene with acetic acid in the presence of a strong acid catalyst.[2]
It can also be produced by reduction of camphor:
Isoborneol derivatives as chiral ligands
Derivatives of isoborneol are used as ligands in asymmetric synthesis.[3]
- (2S)-(−)-3-exo-(morpholino)isoborneol or MIB[4] with a morpholine substituent in the α-hydroxyl position.
- (2S)-(−)-3-exo-(dimethylamino)isoborneol or DAIB[5] with a dimethylamino substituent in the α-hydroxyl position
Remove ads
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads