Top Qs
Timeline
Chat
Perspective
Lactaldehyde
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
Lactaldehyde is an intermediate in the methylglyoxal metabolic pathway. Methylglyoxal is converted to D-lactaldehyde by glycerol dehydrogenase (gldA). Lactaldehyde is then oxidized to lactic acid by aldehyde dehydrogenase.[1]
Remove ads
Structure
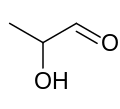
Lactaldehyde is a three-carbon atom species with a carbonyl group on the first carbon atom (making it an aldehyde), and a hydroxy group on the second carbon atom, making it a secondary alcohol. The molecule is chiral, its stereocenter being located on the second carbon atom.
Lactaldehyde exists in several forms: in open-chain form and as cyclic hemiacetal; in solution and in crystal forms; as monomer and as dimer. In crystal form, three conformers occur as hemiacetal dimers with a 1,4-dioxane ring skeleton:

In equilibrium solution, negligibly small amounts of the monomer and at least one five-membered ring dimer exist.[2]
Remove ads
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads