Top Qs
Timeline
Chat
Perspective
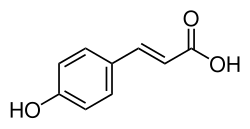
P-Coumaric acid
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
p-Coumaric acid is an organic compound with the formula HOC6H4CH=CHCO2H. It is one of the three isomers of coumaric acid. It is a white solid that is only slightly soluble in water but very soluble in ethanol and diethyl ether.
Remove ads
Natural occurrences
Summarize
Perspective
It is a precursor to many natural products, especially lignols, precursors to the woody mass that comprise many plants.[2] Of the myriad occurrences, p-coumaric acid can be found in Gnetum cleistostachyum.[3]
In food
p-Coumaric acid can be found in a wide variety of edible plants and fungi such as peanuts, navy beans, tomatoes, carrots, basil and garlic.[citation needed] It is found in wine and vinegar.[4] It is also found in barley grain.[5]
Derivatives
p-Coumaric acid glucoside can also be found in commercial breads containing flaxseed.[7] Diesters of p-coumaric acid can be found in carnauba wax.
Biosynthesis
It is biosynthesized from cinnamic acid by the action of the P450-dependent enzyme 4-cinnamic acid hydroxylase (C4H).
It is also produced from L-tyrosine by the action of tyrosine ammonia lyase (TAL).
Biosynthetic building block
p-Coumaric acid is the precursor of 4-ethylphenol produced by the yeast Brettanomyces in wine. The enzyme cinnamate decarboxylase catalyzes the conversion of p-coumaric acid into 4-vinylphenol.[8] Vinyl phenol reductase then catalyzes the reduction of 4-vinylphenol to 4-ethylphenol. Coumaric acid is sometimes added to microbiological media, enabling the positive identification of Brettanomyces by smell.
cis-p-Coumarate glucosyltransferase is an enzyme that uses uridine diphosphate glucose and cis-p-coumarate to produce 4′-O-β-D-glucosyl-cis-p-coumarate and uridine diphosphate (UDP). This enzyme belongs to the family of glycosyltransferases, specifically the hexosyltransferases.[9]
Phloretic acid, found in the rumen of sheep fed with dried grass, is produced by hydrogenation of the 2-propenoic side chain of p-coumaric acid.[10]
The enzyme, resveratrol synthase, also known as stilbene synthase, catalyzes the synthesis of resveratrol ultimately from a tetraketide derived from 4-coumaroyl CoA.[11]
p-Coumaric acid is a cofactor of photoactive yellow proteins (PYP), a homologous group of proteins found in many eubacteria.[12]
p-Coumaric acid is found as the base moiety of caleicine, one of many sesquiterpenes in Calea ternifolia.
Remove ads
See also
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads