Top Qs
Timeline
Chat
Perspective
Praseodymium(III) carbonate
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
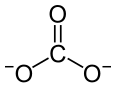
Praseodymium(III) carbonate is an inorganic compound, with a chemical formula of Pr2(CO3)3. The anhydrous form is olive green, and many of its hydrates such as heptahydrate and octahydrate are known.[3] They are all insoluble in water.[2]
Remove ads
Preparation
Praseodymium(III) carbonate can be obtained by the hydrolysis of praseodymium(III) chloroacetate:[2][4]
- 2 Pr(C2Cl3O2)3 + 3 H2O → Pr2(CO3)3 + 6 CHCl3 + 3 CO2
It can also be obtained by reacting sodium bicarbonate saturated with carbon dioxide with a praseodymium chloride solution.[4]
Chemical properties
Praseodymium(III) carbonate is soluble in acids, and emits carbon dioxide:[5]
- Pr2(CO3)3 + 6 H+ → 2 Pr3+ + 3 H2O + 3 CO2↑
However, it is insoluble in water.[2]
Other compounds
Praseodymium(III) carbonate forms compounds with N2H4, such as Pr2(CO3)3•12N2H4•5H2O which is a pale green crystal that is slightly soluble in water but insoluble in benzene, with d20°C = 1.873 g/cm3.[6]
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads