Top Qs
Timeline
Chat
Perspective
Thioformaldehyde
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
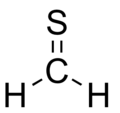
Thioformaldehyde is the organosulfur compound with the formula CH2S. It is the simplest thioaldehyde. This compound is not observed in the condensed state (solid or liquid) because it oligomerizes to 1,3,5-trithiane, which is a stable colorless compound with the same empirical formula.
Despite the instability of these concentrated forms, thioformaldehyde as a dilute gas has been extensively studied. For these purposes, it is produced by thermal decomposition of dimethyl disulfide.[1] The molecule has been observed in the interstellar medium[2] and has attracted much attention for its fundamental nature.[3] The tendency of thioformaldehyde to form chains and rings is a manifestation of the double bond rule.
Although thioformaldehyde tends to oligomerize, many metal complexes are known. One example is Os(SCH2)(CO)2(PPh3)2.[4]


Remove ads
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads