Arsenate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Arsenate?
Summarize this article for a 10 year old
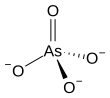
The arsenate is an ion with the chemical formula AsO3−4.[1] Bonding in arsenate consists of a central arsenic atom, with oxidation state +5, double bonded to one oxygen atom and single bonded to a further three oxygen atoms.[2] The four oxygen atoms orient around the arsenic atom in a tetrahedral geometry.[2] Resonance disperses the ion's −3 charge across all four oxygen atoms.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Arsenate | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider |
| ||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| AsO3−4 | |||
| Molar mass | 138.918 g·mol−1 | ||
| Conjugate acid | Arsenic acid | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Extremely toxic, carcinogenic | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Arsenate readily reacts with metals to form arsenate metal compounds.[2][3] Arsenate is a moderate oxidizer and an electron acceptor, with an electrode potential of +0.56 V for its reduction to arsenite.[4] Due to arsenic having the same valency and similar atomic radius to phosphorus, arsenate shares similar geometry and reactivity with phosphate.[5] Arsenate can replace phosphate in biochemical reactions and is toxic to most organisms.[5][6]