
Arsenic trisulfide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Arsenic trisulfide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Arsenic trisulfide is the inorganic compound with the formula As2S3. It is a dark yellow solid that is insoluble in water. It also occurs as the mineral orpiment (Latin: auripigmentum), which has been used as a pigment called King's yellow. It is produced in the analysis of arsenic compounds. It is a group V/VI, intrinsic p-type semiconductor and exhibits photo-induced phase-change properties.[clarification needed]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Arsenic trisulfide | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.013.744 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| As2S3 | |
| Molar mass | 246.02 g·mol−1 |
| Appearance | yellow or orange crystals |
| Density | 3.43 g/cm3 |
| Melting point | 310 °C (590 °F; 583 K) |
| Boiling point | 707 °C (1,305 °F; 980 K) |
| insoluble | |
| Solubility | soluble in ammonia |
| −70.0·10−6 cm3/mol | |
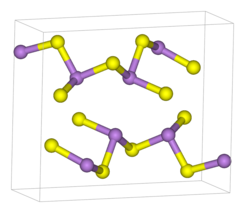
| Structure[1] | |
| monoclinic | |
| P21/n (No. 11) | |
a = 1147.5(5) pm, b = 957.7(4) pm, c = 425.6(2) pm α = 90°, β = 90.68(8)°, γ = 90° | |
| pyramidal (As) | |
| Hazards | |
| GHS labelling:[2][3] | |
  | |
| Danger | |
| H300, H331, H400, H411 | |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
[1910.1018] TWA 0.010 mg/m3[4] |
REL (Recommended) |
Ca C 0.002 mg/m3 [15-minute][4] |
IDLH (Immediate danger) |
Ca [5 mg/m3 (as As)][4] |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
