Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
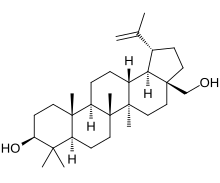
Betulin is an abundant, naturally occurring triterpene. It is commonly isolated from the bark of birch trees. It forms up to 30% of the dry weight of silver birch bark.[2] It is also found in birch sap.[citation needed] Inonotus obliquus contains betulin.[3]
 | |
| Names | |
|---|---|
| IUPAC name
Lup-20(29)-ene-3β,28-diol | |
| Systematic IUPAC name
(1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bR)-3a-(Hydroxymethyl)-5a,5b,8,8,11a-pentamethyl-1-(prop-1-en-2-yl)icosahydro-1H-cyclopenta[a]chrysen-9-ol | |
| Other names
Betulinol, betuline, betulol, betulinic alcohol, trochol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.797 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C30H50O2 | |
| Molar mass | 442.728 g·mol−1 |
| Appearance | solid with needle-like crystals[1] |
| Melting point | 256 to 257 °C (493 to 495 °F; 529 to 530 K) |
| insoluble[1] | |
| Solubility | slightly soluble in ethanol and benzene; soluble in diethyl ether, ethyl acetate and ligroin[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound in the bark gives the tree its white color which appears to protect the tree from mid-winter overheating by the sun. As a result, birches are some of the northernmost occurring deciduous trees.
Betulin was discovered in 1788 by German-Russian chemist Johann Tobias Lowitz.[4][5]
Chemically, betulin is a triterpenoid of lupane structure. It has a pentacyclic ring structure, and hydroxyl groups in positions C3 and C28.
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.