Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
Caesium auride is the inorganic compound with the formula CsAu. It is the Cs+ salt of the unusual Au− anion.[2]
 | |
 | |
 Solution of CsAu(left), pure CsAu(right) | |
| Names | |
|---|---|
| IUPAC name
Caesium auride | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| AuCs | |
| Molar mass | 329.872022 g·mol−1 |
| Appearance | Yellow crystals |
| Melting point | 580 °C (1,076 °F; 853 K)[1] |
| reacts violently | |
| Structure | |
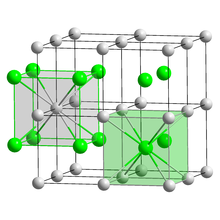
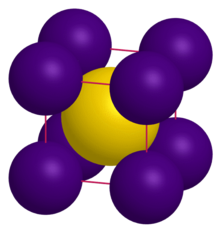
| CsCl | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
CsAu is obtained by heating a stoichiometric mixture of caesium and gold. The two metallic-yellow liquids react to give a transparent yellow product.[3] Despite being a compound of two metals, CsAu lacks metallic properties since it is a salt with localized charges; it instead behaves as a semiconductor with band gap 2.6 eV.[4]
The compound hydrolyzes readily, yielding caesium hydroxide, metallic gold, and hydrogen.[3]
The solution in liquid ammonia is brown, and the ammonia adduct CsAu·NH3 is blue; the latter has ammonia molecules intercalated between layers of the CsAu crystal parallel to the (110) plane. Solutions undergo metathesis with tetramethylammonium loaded ion exchange resin to give tetramethylammonium auride.[3]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.