
Cyclopentadienyliron dicarbonyl dimer
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cyclopentadienyliron dicarbonyl dimer?
Summarize this article for a 10 year old
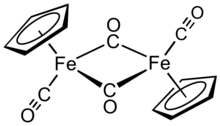
Cyclopentadienyliron dicarbonyl dimer is an organometallic compound with the formula [(η5-C5H5)Fe(CO)2]2, often abbreviated to Cp2Fe2(CO)4, [CpFe(CO)2]2 or even Fp2, with the colloquial name "fip dimer". It is a dark reddish-purple crystalline solid, which is readily soluble in moderately polar organic solvents such as chloroform and pyridine, but less soluble in carbon tetrachloride and carbon disulfide. Cp2Fe2(CO)4 is insoluble in but stable toward water. Cp2Fe2(CO)4 is reasonably stable to storage under air and serves as a convenient starting material for accessing other Fp (CpFe(CO)2) derivatives (described below).[1]
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Di-μ-carbonyldicarbonylbis(η5-cyclopenta-2,4-dien-1-yl)diiron | |
| Other names
Bis(cyclopentadienyl)tetracarbonyl-diiron, Di(cyclopentadienyl)tetracarbonyl-diiron, Bis(dicarbonylcyclopentadienyliron) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.032.057 |
| EC Number |
|
PubChem CID |
|
| |
| |
| Properties | |
| C14H10Fe2O4 | |
| Molar mass | 353.925 g/mol |
| Appearance | Dark purple crystals |
| Density | 1.77 g/cm3, solid |
| Melting point | 194 °C (381 °F; 467 K) |
| Boiling point | decomposition |
| insoluble | |
| Solubility in other solvents | benzene, THF, chlorocarbons |
| Structure | |
| distorted octahedral | |
| 3.1 D (benzene solution) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
CO source |
| GHS labelling: | |
   | |
| Danger | |
| H228, H302, H330, H331 | |
| Related compounds | |
Related compounds |
Fe(C5H5)2 Fe(CO)5 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |