
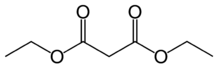
Diethyl malonate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Diethyl malonate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Diethyl malonate, also known as DEM, is the diethyl ester of malonic acid. It occurs naturally in grapes and strawberries as a colourless liquid with an apple-like odour, and is used in perfumes. It is also used to synthesize other compounds such as barbiturates, artificial flavourings, vitamin B1, and vitamin B6.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Diethyl malonate[1] | |
| Preferred IUPAC name
Diethyl propanedioate | |
| Other names
Diethyl malonate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| Abbreviations | DEM |
| 774687 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.003.006 |
| EC Number |
|
| MeSH | Diethyl+malonate |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C7H12O4 | |
| Molar mass | 160.17 g/mol |
| Appearance | colourless liquid |
| Density | 1.05 g/cm3, liquid |
| Melting point | −50 °C (−58 °F; 223 K) |
| Boiling point | 199 °C (390 °F; 472 K) |
| negligible | |
| Acidity (pKa) | 14 16.37 (DMSO)[2] |
| -92.6·10−6 cm3/mol | |
| Structure | |
| 2.54 D (liquid)[3] | |
| Thermochemistry | |
Heat capacity (C) |
285.0 J·mol−1·K−1[3] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Harmful (X), Flammable (F) |
| Flash point | 93 °C (199 °F; 366 K)[3] |
| Safety data sheet (SDS) | Oxford University MSDS |
| Related compounds | |
Related compounds |
Dimethyl malonate Malonic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close