
Etravirine
Also called Intelence is a drug used for the treatment of HIV / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Etravirine?
Summarize this article for a 10 year old
Etravirine (ETR,[1] brand name Intelence, formerly known as TMC125) is a drug used for the treatment of HIV. Etravirine is a non-nucleoside reverse transcriptase inhibitor (NNRTI). Unlike the currently available agents in the class, resistance to other NNRTIs does not seem to confer resistance to etravirine.[2] Etravirine is marketed by Janssen, a subsidiary of Johnson & Johnson. In January 2008, the Food and Drug Administration approved its use for patients with established resistance to other drugs, making it the 30th anti-HIV drug approved in the United States and the first to be approved in 2008.[3] It was also approved for use in Canada on April 1, 2008.[4]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Intelence |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608016 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 99.9% |
| Metabolism | Hepatic (CYP3A4, CYP2C9 & CYP2C19-mediated) |
| Elimination half-life | 41±20 hours |
| Excretion | Faeces (93.7%), urine (1.2%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| NIAID ChemDB | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.207.546 |
| Chemical and physical data | |
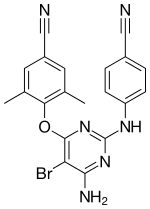
| Formula | C20H15BrN6O |
| Molar mass | 435.285 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Etravirine is licensed in the United States, Canada, Israel, Russia, Australia, New Zealand and the European Union,[5] and is under regulatory review in Switzerland.[6]