
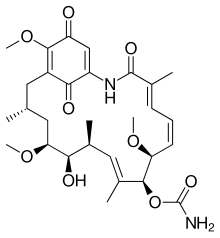
Geldanamycin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Geldanamycin?
Summarize this article for a 10 year old
Geldanamycin is a 1,4-benzoquinone ansamycin antitumor antibiotic that inhibits the function of Hsp90 (Heat Shock Protein 90) by binding to the unusual ADP/ATP-binding pocket of the protein.[1] HSP90 client proteins play important roles in the regulation of the cell cycle, cell growth, cell survival, apoptosis, angiogenesis and oncogenesis.[2]

 | |
| Names | |
|---|---|
| IUPAC name
(4E,6Z,8S,9S,10E,12S,13R,14S,16R)-13-hydroxy- | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C29H40N2O9 | |
| Molar mass | 560.64 g/mol |
| Appearance | Gold-yellow fine crystalline powder |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Geldanamycin induces the degradation of proteins that are mutated or overexpressed in tumor cells such as v-Src, Bcr-Abl, p53, and ERBB2. This effect is mediated via HSP90. Despite its potent antitumor potential, geldanamycin presents several major drawbacks as a drug candidate such as hepatotoxicity, further, Jilani et al.. reported that geldanamycin induces the apoptosis of erythrocytes under physiological concentrations.[4] These side effects have led to the development of geldanamycin analogues, in particular analogues containing a derivatisation at the 17 position: