
Lithium sulfate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lithium sulfate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Lithium sulfate is a white inorganic salt with the formula Li2SO4. It is the lithium salt of sulfuric acid.
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Lithium sulfate | |
| Other names
Lithium sulphate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.030.734 |
PubChem CID |
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties[1] | |
| Li2SO4 | |
| Molar mass | 109.94 g/mol |
| Appearance | White crystalline solid, hygroscopic |
| Density | 2.221 g/cm3 (anhydrous) 2.06 g/cm3 (monohydrate) |
| Melting point | 859 °C (1,578 °F; 1,132 K) |
| Boiling point | 1,377 °C (2,511 °F; 1,650 K) |
| monohydrate: 34.9 g/100 mL (25 °C) 29.2 g/100 mL (100 °C) | |
| Solubility | insoluble in absolute ethanol, acetone and pyridine |
| −-40.0·10−6 cm3/mol | |
Refractive index (nD) |
1.465 (β-form) |
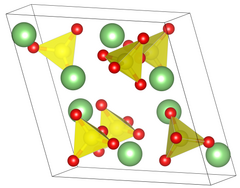
| Structure[2] | |
| Primitive monoclinic | |
| P 21/a, No. 14 | |
a = 8.239 Å, b = 4.954 Å, c = 8.474 Å | |
Lattice volume (V) |
328.9 Å3 |
Formula units (Z) |
4 |
| Tetrahedral at sulfur | |
| Thermochemistry | |
Heat capacity (C) |
1.07 J/g K |
Std molar entropy (S⦵298) |
113 J/mol K |
Std enthalpy of formation (ΔfH⦵298) |
−1436.37 kJ/mol |
Gibbs free energy (ΔfG⦵) |
-1324.7 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
613 mg/kg (rat, oral)[3] |
| Related compounds | |
Other anions |
Lithium chloride |
Other cations |
Sodium sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
