
Magic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Magic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Magic acid (FSO3H·SbF5) is a superacid consisting of a mixture, most commonly in a 1:1 molar ratio, of fluorosulfuric acid (HSO3F) and antimony pentafluoride (SbF5). This conjugate Brønsted–Lewis superacid system was developed in the 1960s by the George Olah lab at Case Western Reserve University, and has been used to stabilize carbocations and hypercoordinated carbonium ions in liquid media. Magic acid and other superacids are also used to catalyze isomerization of saturated hydrocarbons, and have been shown to protonate even weak bases, including methane, xenon, halogens, and molecular hydrogen.[1]
Quick Facts Names, Identifiers ...
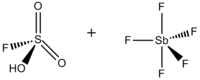 Fluorosulfuric acid-antimony pentafluoride 1:1 | |
 | |
| Names | |
|---|---|
| IUPAC name
Sulfurofluoridic acid — pentafluorostiborane (1:1) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.041.727 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| HSbF6SO3 | |
| Molar mass | 316.82 g/mol |
| Appearance | Liquid |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Extremely corrosive, toxic, violent hydrolysis, oxidizer. |
| GHS labelling: | |
      | |
| Danger | |
| H240, H271, H290, H302, H314, H335, H350, H402, H410, H411, H441 | |
| P234, P261, P264, P270, P271, P273, P280, P284, P301+P310, P301+P330+P331, P303+P361+P353, P304+P340+P310, P305+P351+P338+P310, P363, P390, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
