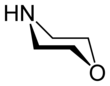
Morpholine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Morpholine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with morphine or morpholino.
Morpholine is an organic chemical compound having the chemical formula O(CH2CH2)2NH. This heterocycle features both amine and ether functional groups. Because of the amine, morpholine is a base; its conjugate acid is called morpholinium. For example, treating morpholine with hydrochloric acid makes the salt morpholinium chloride. It is a colorless liquid with a weak, ammonia- or fish-like odor.[6] The naming of morpholine is attributed to Ludwig Knorr, who incorrectly believed it to be part of the structure of morphine .[7]
Quick Facts Names, Identifiers ...
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Morpholine[2] | |||
| Other names
Diethylenimide oxide 1,4-Oxazinane Tetrahydro-1,4-oxazine Diethylene imidoxide Diethylene oximide Tetrahydro-p-oxazine | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| 102549 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.003.469 | ||
| EC Number |
| ||
| 1803 | |||
| KEGG |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2054 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C4H9NO | |||
| Molar mass | 87.122 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Odor | Weak ammonia-like or fish-like[3] | ||
| Density | 1.007 g/cm3 | ||
| Melting point | −5 °C (23 °F; 268 K) | ||
| Boiling point | 129 °C (264 °F; 402 K) | ||
| miscible | |||
| Vapor pressure | 6 mmHg (20 °C)[3] | ||
| Acidity (pKa) | 8.36[4] (of conjugate acid) | ||
| -55.0·10−6 cm3/mol | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Flammable, Corrosive | ||
| GHS labelling: | |||
   | |||
| Danger | |||
| H226, H302, H312, H314, H332 | |||
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P370+P378, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 31 °C (88 °F; 304 K) | ||
| 275 °C (527 °F; 548 K) | |||
| Explosive limits | 1.4%–11.2%[3] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
1220 mg/kg (mammal, oral) 525 mg/kg (mouse, oral) 1050 mg/kg (rat, oral)[5] | ||
LC50 (median concentration) |
365 ppm (mouse, 2 hr)[5] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 20 ppm (70 mg/m3) [skin][3] | ||
REL (Recommended) |
TWA 20 ppm (70 mg/m3) ST 30 ppm (105 mg/m3) [skin][3] | ||
IDLH (Immediate danger) |
1400 ppm[3] | ||
| Safety data sheet (SDS) | hazard.com | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close