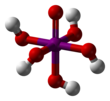
Periodic acid
Oxoacid of iodine (H5IO6 or HIO4) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Periodic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Periodic acid (/ˌpɜːraɪˈɒdɪk/ per-eye-OD-ik) is the highest oxoacid of iodine, in which the iodine exists in oxidation state +7. It can exist in two forms: orthoperiodic acid, with the chemical formula H5IO6, and metaperiodic acid, which has the formula HIO4.
Quick Facts Names, Identifiers ...
Orthoperiodic acid | |||
Metaperiodic acid | |||
 Orthoperiodic acid | |||
| Names | |||
|---|---|---|---|
Other names
| |||
| Identifiers | |||
| |||
3D model (JSmol) |
| ||
| ChemSpider | |||
| ECHA InfoCard | 100.030.839 | ||
| EC Number |
| ||
PubChem CID |
| ||
| UNII |
| ||
| UN number | UN3085 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| HIO4 (metaperiodic) H5IO6 (orthoperiodic) | |||
| Molar mass | 190.91 g/mol (HIO4) 227.941 g/mol (H5IO6) | ||
| Appearance | Colourless crystals | ||
| Melting point | 128.5 °C (263.3 °F; 401.6 K)[1] | ||
| Solubility | soluble in water, alcohols | ||
| Conjugate base | Periodate | ||
| Hazards[2] | |||
| GHS labelling: | |||
    | |||
| Danger | |||
| H271, H314, H372, H400 | |||
| P210, P260, P273, P303+P361+P353, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Other anions |
| ||
Other cations |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Periodic acid was discovered by Heinrich Gustav Magnus and C. F. Ammermüller in 1833.[3]