
Potassium ferrate
Chemical compound / From Wikipedia, the free encyclopedia
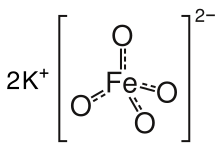
Potassium ferrate is the chemical compound with the formula K2FeO4. This purple salt is paramagnetic, and is a rare example of an iron(VI) compound. In most of its compounds, iron has the oxidation state +2 or +3 (Fe2+ or Fe3+). Reflecting its high oxidation state, FeO2−4 is a powerful oxidizing agent.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Potassium ferrate(VI) | |
| Other names
Potassium ferrate Dipotassium ferrate | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| K2FeO4 | |
| Molar mass | 198.0392 g/mol |
| Appearance | Dark purple solid |
| Density | 2.829 g/cm3 |
| Melting point | >198 °C (decomposes) |
| soluble in 1M KOH | |
| Solubility in other solvents[which?] | reacts with most solvents |
| Structure | |
| K2SO4 motif | |
| Tetrahedral | |
| 0 D | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Oxidizer |
| GHS labelling: | |
 [1] [1] | |
| Danger[1] | |
| H272[1] | |
| P210, P220, P221, P280, P370+P378, P501[1] | |
| Flash point | non-combustible |
| Safety data sheet (SDS) | External SDS |
| Related compounds | |
Other anions |
K2MnO4 K2CrO4 K2RuO4 |
Other cations |
BaFeO4 Na2FeO4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close