
Pyocyanin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Pyocyanin?
Summarize this article for a 10 year old
Pyocyanin (PCN−) is one of the many toxic compounds produced and secreted by the Gram negative bacterium Pseudomonas aeruginosa. Pyocyanin is a blue secondary metabolite, turning red below pH 4.9, with the ability to oxidise and reduce other molecules[2] and therefore kill microbes competing against P. aeruginosa as well as mammalian cells of the lungs which P. aeruginosa has infected during cystic fibrosis.[3][4] Since pyocyanin is a zwitterion at blood pH, it is easily able to cross the cell membrane. There are three different states in which pyocyanin can exist: oxidized (blue), monovalently reduced (colourless) or divalently reduced (red). Mitochondria play an important role in the cycling of pyocyanin between its redox states. Due to its redox-active properties, pyocyanin generates reactive oxygen species.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
5-Methylphenazin-1(5H)-one | |
| Other names
Pyocyanin; Pyrocyanine; 5-Methyl-1(5H)-phenazinone; Sanasin; Sanazin | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.213.248 |
| EC Number |
|
| KEGG | |
| MeSH | D011710 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
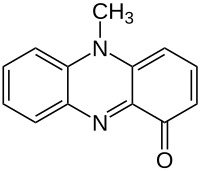
| C13H10N2O | |
| Molar mass | 210.236 g·mol−1 |
| Appearance | Solid |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H302, H318 | |
| P264, P270, P280, P301+P312, P305+P351+P338, P310, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |