
Thallium(I) sulfate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Thallium(I) sulfate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Thallium(I) sulfate (Tl2SO4) or thallous sulfate is the sulfate salt of thallium in the common +1 oxidation state, as indicated by the Roman numeral I. It is often referred to as simply thallium sulfate.[2]
Quick Facts Names, Identifiers ...
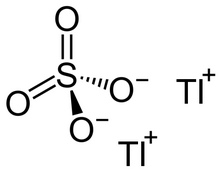 | |
| Names | |
|---|---|
| Other names
Thallous sulfate, Thallium sulfate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.365 |
| KEGG |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1707 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Tl2SO4 | |
| Molar mass | 504.83 g/mol |
| Appearance | white prisms or dense white powder |
| Odor | odorless |
| Density | 6.77 g/cm3 |
| Melting point | 632 °C (1,170 °F; 905 K) |
| 2.70 g/100 mL (0 °C) 4.87 g/100 mL (20 °C) 18.45 g/100 mL (100 °C) | |
| −112.6·10−6 cm3/mol | |
Refractive index (nD) |
1.860 |
| Structure | |
| rhomboid | |
| Hazards | |
| GHS labelling: | |
    | |
| Danger | |
| H300, H315, H372, H411 | |
| P260, P264, P270, P273, P280, P301+P310, P302+P352, P314, P321, P330, P332+P313, P362, P391, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
16 mg/kg (rat, oral) 23.5 mg/kg (mouse, oral)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
