
Trithioacetone
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trithioacetone?
Summarize this article for a 10 year old
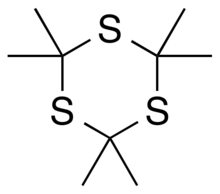
Trithioacetone (2,2,4,4,6,6-hexamethyl-1,3,5-trithiane) is an organic chemical with formula C
9H
18S
3. Its covalent structure is [–C(CH
3)
2–S–]
3, that is, a six-membered ring of alternating carbon and sulfur atoms, with two methyl groups attached to each carbon.[4][2] It can be viewed as a derivative of 1,3,5-trithiane, with methyl-group substituents for all of the hydrogen atoms in that parent structure.
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Hexamethyl-1,3,5-trithiane | |
| Other names
Trithioacetone[1] | |
| Identifiers | |
3D model (JSmol) |
|
| 5-19-09-00119 | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.011.438 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H18S3 | |
| Molar mass | 222.42 g·mol−1 |
| Density | 1.0660 to 1.0700 g/mL[2] |
| Melting point | 21.8°C[3] |
| Boiling point | 107°C/10mmHg[2] |
Refractive index (nD) |
1.5390 to 1.5430[2] |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound Trithioacetone is a stable cyclic trimer of thioacetone (propane-2-thione), which by itself is an unstable compound.[5][6] In contrast, the analogous trioxane compound, 2,2,4,4,6,6-hexamethyl-1,3,5-trioxane (Triacetone), with oxygen atoms in place of the sulfur atoms, seems to be unstable, while its corresponding monomer acetone (2-propanone) is stable.